Difference between revisions of "Kidney tumours"
m (→IHC) |
(split-out) |
||
| Line 464: | Line 464: | ||
=Renal translocation carcinomas= | =Renal translocation carcinomas= | ||
==Renal tumour with Xp11.2 translocation== | ==Renal tumour with Xp11.2 translocation== | ||
{{Main|Renal tumour with Xp11.2 translocation}} | |||
==Renal tumour with t(6;11) translocation== | ==Renal tumour with t(6;11) translocation== | ||
Revision as of 16:16, 23 November 2013
Kidney tumours - includes malignant kidney tumours and benign kidney tumours. Medical renal diseases are dealt with in the medical renal diseases article.
Pediatric kidney tumours are dealt with in the pediatric kidney tumours article.
Renal specimens
- Renal biopsy - usually for renal onocytoma vs. renal cell carcinoma (RCC) or medical diseases - see medical kidney.
- Partial nephrectomy.
- Nephrectomy.
- Nephroureterectomy (includes ureter) - done for urothelial cell carcinoma (UCC) of the renal pelvis and ureteric UCC.
- Radical nephrectomy - includes Gerota's fascia.
- May include the adrenal gland.[1]
In excisions done for tumours, a comment should be made about kidney distant from the tumour. People with less renal mass, i.e. less kidney, are predisposed to focal segmental glomerulosclerosis (FSGS).
Anatomy
The anatomy is important for properly staging renal neoplasms.
Layers (superficial to deep):
- Renal fascia (Gerota's fascia).
- Perinephric fat.
- Renal capsule.
- Renal parenchyma (cortex).
Sign out
Missed renal biopsy
KIDNEY (LESION), LEFT, CORE BIOPSY: - RENAL PARENCHYMA. - NEGATIVE FOR MASS LESION, SEE COMMENT. COMMENT: No mass lesion is apparent in the tissue sampled. A re-biopsy should be considered. Renal parenchyma: - Glomeruli: seven glomeruli sampled, no apparent glomerular pathology on the H&E sections. - Interstitium: interstitial fibrosis is not identified. - Tubules: no pathology is apparent. - Vessels: mild atherosclerosis, no hyalinization of arterioles apparent.
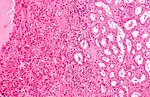
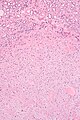
Tabular comparison (selected tumours)
Selected common tumours of the kidney:[2][3]
| Clear cell RCC | Papillary RCC type 1 |
Papillary RCC type 2 |
Chromophobe RCC classic variant |
Chromophobe RCC eosinophilic variant |
Oncocytoma | |
| Gross | Golden yellow, solid | friable | friable | light brown | light brown | mahogany/brown, +/-central scar |
| Architecture | nests, sheets | papillary, simple | papillary, pseudostratified |
nests, sheets | nests, sheets | nests, sheets |
| Nuclear atypia | low-high typically medium-high |
low-medium | medium-high | low-high, "raisinoid" nuc. membrane |
low-high, "raisinoid" nuc. membrane |
low-medium, round nuclei |
| Cytoplasm | clear | eosinophilic | eosinophilic | cobwebs/clear | eosinophilic/cobwebs | eosinophilic/ granular & abundant |
| Other | delicate vessels, necrosis common |
histiocytes in fibrovascular cores, >0.5 cm |
histiocytes in fibrovascular cores, >0.5 cm |
perinuclear clearing, thick vessels | perinuclear clearing, thick vessels | in loose fibrous/hyaline stroma |
| IHC | CK7-, EMA+ | AMACR+, EMA+, CK7+ | AMACR+, E-cadherin+, CK7- | CD117+, CK7+ (membrane) | CD117+, CK7+ (membrane) | Vimentin-, EMA+ |
| Main DDx | chromophobe classic variant |
PaRCC type 2, mets | PaRCC type 1, mets | clear cell RCC | oncocytoma | chromophobe eosinophilic variant |
| Key features | clear cells, vascular | papillae, histiocytes simple epithelium |
papillae, histiocytes, stratified |
perinuc. clearing, wispy cytoplasm |
perinuc. clearing, wispy eosinophilic cytoplasm |
eosinophilic, granular cytoplasm |
| Image(s) | , |
Notes:
- Cell shape: all have epithelioid morphology.
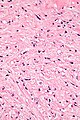
Tabular comparison of oncocytoma and chromophobe RCC
Histomorphologic features useful to distinguish chromophobe RCC (eosinophilic variant) and oncocytoma:[4]
| Morphologic feature | ChRCC (eosinophilic variant) |
Renal oncocytoma |
|---|---|---|
| Nuclear morphology | "raisinoid"/wrinkled appearance | round with small nucleolus, usu. little size variation |
| Multinucleation | common - binucleation | uncommon |
| Chromatin | coarse | fine |
| Architecture | solid, crowded nests | spaced nests / archipelago-like, solid |
| Cytoplasm | perinuclear halo, may be focal | no perinuclear halo |
| Degenerative foci (focal atypia & pleomorphism) |
absent | present in ~20% of cases |
| Image |
Common DDx
Spindle cell tumours:
- Malignant:
- Benign:
WHO classification
- Based on 2004 iteration - as per WMSP, slightly modified.[5] Online, the classification can found here.
Renal cell tumours
Common:
- Clear cell renal cell carcinoma.
- Multilocular clear cell renal cell carcinoma.
- Papillary renal cell carcinoma.
- Papillary adenoma.
- Chromophobe renal cell carinoma.
- Oncocytoma.
Less common:
- Carcinoma of the collecting ducts of Bellini.
- Renal cell carcinoma, unclassified.
- Renal medullary carcinoma.
- Xp11 translocation carcinoma.
- Mucinous tubular and spindle cell carcinoma.
- Carcinoma associated with neuroblastoma.
Metanephric tumours
Nephroblastic tumours
- Nephrogenic rests.
- Nephroblastoma.
Mesenchymal tumours
Childhood:
Adults:
- Unique to kidney:
Other:
- Angiomyolipoma.
- Epithelioid angiomyolipoma.
- Leiomyosarcoma.
- Angiosarcoma.
- Pleomorphic undifferentiated sarcoma.
- Hemangiopericytoma.
- Solitary fibrous tumour.
- Osteosarcoma.
- Schwannoma.
Mixed mesenchymal and epithelial tumours
Others
- Neuroendocrine tumours.
- Hematologic tumours.
- Germ cell tumours.
- Metastases.
Renal cell carcinoma
Overview
General
- Relatively common form of cancer.
- Often abbreviated RCC.
- AKA hypernephroma.[6]
- RCC represents approx. 90% of malignancies in kidneys of adults.[7]
Origin
- Proximal renal tubule.
Clinical
- Classically described as a triad:[8]
- Hematuria (most common symptom).
- Abdominal mass.
- Flank pain.
- Frequently picked-up on imaging (incidentaloma) ~ 1/3 of cases.
Risk factors
- Smoking - most important.[7]
- Chemical exposures (arsenic, asbestos, cadmium, organic solvents, pesticides, fungal toxins).[7]
- Chronic renal failure.
- Male>female (~2:1).
- Hereditary - familial syndromes (see Hereditary RCC).
Subtypes of RCC
RCC (renal cell carcinoma) comes in different subtypes:[9]
- Clear cell carcinoma (70-80% of RCC) -- abbrev. CCRCC,
- Papillary renal cell carcinoma (10-15% of RCC) -- abbrev. PRCC,
- Chromophobe renal carcinoma (5% of RCC) -- abbrev. ChRCC,
- Collecting duct (Bellini duct) carcinoma (1% of RCC).
Notes:
- Subtype is an independent predictor of mortality - but adds very little to multivariate models with staging information.[10]
- CCRCC tends to be worse than ChRCC and PRCC, probably due to higher incidence of mets.[11]
- The exam answer (worst to best): clear cell RCC, papillary RCC, chromophobe RCC.
IHC - is it RCC?
- RCC Ma (+), CD10 (+) -- specific for RCC[12]
IHC - differentiation of types
- Clear cell RCC vs. papillary RCC:
- CK7 (-ve CCRCC), AMACR (+ve in PRCC).[13]
- Papillary RCC type 1 vs. papillary RCC type 2:
- ChRCC vs. oncocytoma (ONC):
- CK7 (ChRCC +ve membrane), CK20, CD15.[13]
- CK7 -- ChRCC 86% +ve vs. ONC 0% +ve.[15]
- CD15 -- ChRCC 11% +ve vs. ONC 57% +ve.[16]
- Hale's colloidal iron +ve in ChRCC, usually neg. in ONC.[17]
- PAX2 -- ChRCC (1/11) +ve vs. ONC (20/23) +ve.[18]
- Kidney-specific cadherin (Ksp-cadherin) -- ChRCC 97% +ve (distinctive membrane pattern) vs. ONC only 3% +ve.[19]
- ChRCC & renal oncocytoma vs. others:
- CD117 (ckit) +ve (100% membrane, ~75% cytoplasmic).[20]
- Clear cell RCC vs. chromophobe RCC:
- Hale's colloidal iron (+ve in ChRCC).[17]
- CK7 (cell membrane +ve in ChRCC).
Notes:
- One paper[21] describes CD10, parvalbumin, AMACR, CK7 and S100A1 as being useful.
- Another paper I came across:[22]
- c-kit (CD117) not useful for differentiating ONC and ChRCC.[18]
- E-cadherin not useful for differentiating ChRCC and ONC.[23]
RCC vs. Urothelial cell carcinoma
- Clinically/radiologically, it may not be possible to differentiate renal pelvis UCC and RCC if the tumour is large.
- Pathologically, this is not very difficult.
- On gross specimens, it is almost always obvious what one is dealing with:
Renal cell carcinoma with sarcomatoid differentiation
- AKA sarcomatoid renal cell carcinoma.
General
Features:[24]
- Not recognized as a distinct entity in 2004 WHO classification.[25]
- Tend to present at higher stage.
- Worse prognosis when adjusted for stage.
- Fuhrman grade 4 by definition.
Microscopic
Features:[24]
- Renal cell carcinoma.
- Sarcomatoid component:
- Fibrosarcoma - most common.
- Undifferentiated - common.
- Rhabdomyosarcoma - very rare.
Note:
- In essence, any kidney tumour with spindle cells[27] or rhabdoid cells should make one think of this.
Images:
- Sarcomatoid change in RCC (webpathology.com).
- Sarcomatoid changes in CCRCC (ascopubs.org).[28]
- Sarcomatoid changes in RCC (scielo.br).[27]
Hereditary renal cell carcinoma
The classics - which are all autosomal dominant:[9]
- Von Hippel-Lindau syndrome.
- VHL gene mutation.
- Clear cell RCC.
- Hereditary clear cell renal cell carcinoma.
- VHL gene mutation.
- Hereditary papillary renal cell carcinoma.
- MET proto-oncogene mutation.
- PaRCC type 1.[29]
- Hereditary leiomyomatosis and renal cell cancer:[29]
- FH (fumarate hydratase) gene mutation.[30]
- PaRCC type 2.
- Benign leiomyomas skin/uterus.
- Uterine leiomyosarcoma.
- Birt–Hogg–Dubé syndrome:[29]
- FLCN (folliculin) gene mutation.[31]
- Skin lesions (fibrofolliculoma, trichodiscoma, acrochordon).
- ChRCC most common, other types seen (e.g. oncocytoma).
- Variable penetrance (autosomal dominant).
Others:
- Hereditary papillary carcinoma (TFE3 related translocations).[32]
Notes:
- A total of ten hereditary renal cancer syndromes have been described. In eight of the ten the gene is known.[33]
Molecular
Recurrent molecular changes in RCC:
- Clear cell RCC:
- Loss of 3p - contains the VHL gene.
- Papillary RCC:
- Sporadic:
- Trisomy 7, 16, 17.
- Loss of Y.
- Familial:
- Trisomy 7 - contains MET gene.[34]
- Sporadic:
Renal cell carcinoma grading
- AKA Fuhrman grade.
General
Some RCC subtypes are graded based on the Fuhrman system which considers:[35]
- Nuclear pleomorphism (size, shape).
- Chromatin pattern.
- Nucleoli prominence.
Notes:
- The system was validated for clear cell RCC.
- Fuhrman nuclear grade is not prognostic in chromophobe RCC and should not be used in that context.[36]
- Anything with spindle cells[27] or rhabdoid cells should make one think grade 4, as it is likely a renal cell carcinoma with sarcomatoid differentiation.
Criteria & grades
- Grade 1: no nucleoli, near 'normal' appearance.
- Grade 2: finely granular chromatin (key feature), no nuclei visible with 10x objective lens.
- Grade 3: nucleoli seen easily (key feature).
- Grade 4: prominent pleomorphism (key feature), hyperchromasia, macronucleoli.
- Spindle cells[27] or rhabdoid cells also make something grade 4 (see renal cell carcinoma with sarcomatoid differentiation).
Note:
- Most tumours are grade 2 & 3.
Fuhrman grading in short
- 1 vs. 2: grade 2 has granular chromatin, grade 2 has nucleoli visible @ 20x objective.[37]
- 2 vs. 3: grade 3 has nucleoli @ 10x objective.
- 3 vs. 4: grade 4 has pleomorphism/hyperchromasia (or spindle cells or rhabdoid cells).
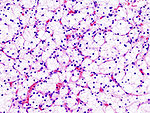
Clear cell renal cell carcinoma
Multilocular cystic renal cell carcinoma
General
- No recurrences or metastasis in the literature.[38]
- This makes one wonder... is it really cancer.
- Case report rare.[39]
Gross
Features:[38]
- Cystic with thin septa.
- Well circumscribed.
Note:
- This tumour, radiologically, can often be separated from other cystic tumours.[40]
Microscopic
Features:[38]
- Polygonal cells within the septa.
- Clear cytoplasm.
- +/-Calcification (common).
DDx:
- Cystic renal disease with macrophages in the septa.
- Cystic clear cell renal cell carcinoma.
- Tubulocystic carcinoma.
IHC
- EMA +ve.
- Keratins +ve.
- CD68 -ve.
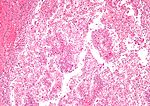
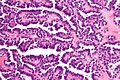
Papillary renal cell carcinoma
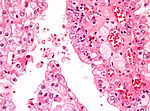
Chromophobe renal cell carcinoma
Clear cell papillary renal cell carcinoma
Unclassified renal cell carcinoma
- Abbreviated URCC.
General
- Uncommon.
- A WHO classification diagnosis of exclusion.
- Worse prognosis than clear cell renal cell carcinoma.[41]
- High variation in the prevalence (when comparing institutions); this suggests a lack of uniformity in the diagnosis of this subtype.[41]
Microscopic
Features:
- Malignant tumour that is one of the following:[42][43]
- A combination of other RCC histologic types (~35% of cases of URCC).
- Has a "non-identifiable" pattern/unrecognizable cell type (~60% of cases of URCC).
- Pure sarcomatoid RCC without an identifiable (epithelioid) RCC subtype (~5% of cases of URCC).[43]
DDx:
- Clear cell renal cell carcinoma.
- Collecting duct carcinoma.
- Undifferentiated carcinoma.
- Metastatic carcinoma.
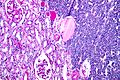
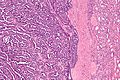
Renal translocation carcinomas
Renal tumour with Xp11.2 translocation
Renal tumour with t(6;11) translocation
General
- Not common.
- Lymph node metastases are common.
- Essentially a pediatric tumour - case report of an adult.[44]
Microscopic
Features:
- Hyaline material between nests.
- Large cells with clear to eosinophilic cytoplasm.
Molecular
- t(6;11)(p21;q12) Alpha/TFEB.[45]
Benign tumours
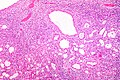
Papillary adenoma of the kidney
- AKA renal papillary adenoma.
General
- Benign.
- Considered a precursor for papillary renal cell carcinoma (PaRCC).[46]
- Stains like PaRCC.
- Found with an increased frequency within the content of PaRCC.
Microscopic
Features:
- Histomorphology of papillary renal cell carcinoma.
- Must be <=0.5 cm.[47][48]
- Diagnostic size cutoff - larger lesions are papillary renal cell carcinoma.
DDx:
Images:
IHC
- AMACR +ve.[46]
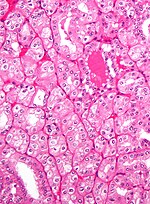
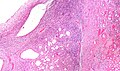
Renal oncocytoma
Angiomyolipoma
- Abbreviated AML.
General
- Benign mesenchymal tumour.
- Presentations: flank pain, hematuria, incidentaloma.[49]
- Tumours >4 cm considered a risk for bleeding.[50]
- AMLs occur may be elsewhere in the body, e.g. liver,[51] but are most common in the kidney.
- In the PEComa group of tumours.
Epidemiology
- May be associated with tuberous sclerosis -- 70% have an AML.
- When compared to sporadic cases:
- More often bilateral.
- Usually bigger.
- When compared to sporadic cases:
- There is a suggestion that an epithelioid variant is more worisome.[52]
- This is not confirmed by all studies.[53]
Microscopic
Features:
- Smooth muscle.
- Adipose tissue - not always present[54] - key feature.
- Abundant blood vessels.
Epithelioid angiomyolipoma
Features:
- Carcinoma-like morphology.
- +/-Spindle cells.
- "High grade" nuclei.
- Pleomorphic nuclei.
DDx:
- Clear cell renal cell carcinoma eosinophilic variant - esp. if epithelioid.
Images:
- Epithelioid AML (birjournals.org).
- Epithelioid AML (rsna.org).
- Atypical epithelioid AML (archivesofpathology.org).[55]
Cytologic
Features[54]
- Nuclei - round/ovoid.
- Chromatin - bland.
IHC
- Ki-67:[57]
- Epithelioid variant of AML +ve.
- Conventional AML -ve.
Mimics
Xanthogranulomatous pyelonephritis
General
- May mimic RCC - espically radiologically.
- Usually lower pole.[citation needed]
- Associated with:
- Diabetes mellitus.
- History of UTI.[58]
- Nephrolithiasis.
- GU obstruction.[59]
- Occasionally an indication of nephrectomy.[58][59]
- Most common organism (in the context of nephrectomy specimens) - Proteus mirabilis.[59]
Microscopic
- Abundant macrophages.
- +/-Giant cells.
DDx:
- Malakoplakia.
- Basophilic inclusions -- inside or outside of macrophages - often size of RBC or larger (Michaelis-Gutmann bodies).
- RCC - especially PRCC (as this has foamy macrophages).
- Granulomatous disease.
- Chronic pyelonephritis.
- Interstitial nephritis.
Image
Stains
- PAS-D -ve.
- Done to look for malakoplakia.
IHC
- CD68 +ve.
- RCC markers (CD10, RCC) all negative.
Malakoplakia
Rare stuffs
Juxtaglomerular cell tumour
General
- Rare.
- Etiology: increased renin.
Clinical:[60]
- Hypertension.
- Increased aldosterone.
- Causes hypokalemia.
- Increased plasma renin.
Microscopic
Features:[61]
- Polygonal cells.
- Abundant granular, eosinophilic cytoplasm.[62]
- Perinuclear halo.
DDx:
- Chromophobe renal cell carcinoma, eosinophilic variant.
Image:
Stains
Cytoplasmic granules.[62]
- PAS +ve.
- PASD +ve.
IHC
Features:[61]
- Actin +ve.
- Cytokeratin -ve.
- HMB-45 -ve.
EM
Features:
- Vesicles - contain renin.[63]
Renomedullary interstitial cell tumour
General
- Benign.
- Common autopsy finding[65] - one review says 26-41% of individuals at autopsy.[66]
- The commonality is somewhat in dispute.[67]
Gross
- Small, white well-circumscribed nodule in medulla.
- Typically less than 3 mm.[66]
Image:
Microscopic
- Small polygonal/stellate cells.
- Abundant loose/myxoid stroma.
- +/-Entrapped renal tubules.[69]
Images
www:
- Renomedullary interstitial cell tumour - low mag. (webpathology.com).
- Renomedullary interstitial cell tumour - high mag. (webpathology.com).
Metanephric adenoma
- Should not be confused mesonephric adenoma, another term for nephrogenic adenoma.
- Memory device: metanephric adenoma is a tumour.
General
- Benign.
- Afflicts adults and occasionally children.
- May be associated with polycythemia.[70]
Microscopic
Features:[71]
- Small uniform cells with:
- Fine chromatin.
- No apparent nucleolus.
- A relatively smooth nuclear membrane.
- Variable architecture - may be sheets or ductal.
DDx:
- Epithelioid nephroblastoma (Wilms tumour) - these typically have:
- Irregular nuclear membrane.
- Nucleoli.
- Mitoses (rare in metanephric adenoma).
- Papillary RCC.[71]
Images
www:
IHC
Renal epithelial and stromal tumour
- Abbreviated REST.
The lumping term for both:[73]
Mixed epithelial and stromal tumour
- Abbreviated MEST.
General
- Rare - approx. 1.5% of renal neoplasms.[74]
- Benign.
- Prevalence: males > females.
Note:
- Turbiner et al.[73] have suggested that cystic nephroma and mixed epithelial and stromal tumour (MEST) are one tumour.
- The lumping term renal epithelial and stromal tumour, abbreviated REST.
Microscopic
Features:
- Cysts lined by simple epithelium with hobnailing - key feature.
- Stroma has an ovarian look:
- Blue (basophilic).
- Spindle cells.
Notes:
- Parenchymal elements (e.g. glomueruli, tubules) are not found in the septa between the cysts.
Images
IHC
Features:[74]
- ER +ve.
- PR +ve.
- CD10 +ve.
DDx
- Tubulocystic carcinoma.
Cystic nephroma
General
- Turbiner et al.[73] has suggested that cystic nephroma and mixed epithelial and stromal tumour (MEST) are one tumour.
- The lumping term is renal epithelial and stromal tumour, abbreviated REST.
Gross
- Bubble wrap-like appearance.
Image
Microscopic
Features:
- Cysts lined by simple epithelium with hobnailing - key feature.
- Stroma has an ovarian look:
- Blue (basophilic).
- Spindle cells.
Notes:
- Parenchymal elements (e.g. glomueruli, tubules) are not found in the septa between the cysts.
Images
IHC
Features:
- ER +ve.
- PR +ve.
- CD10 +ve.
Renal mucinous tubular and spindle cell carcinoma
- AKA renal mucinous tubular spindle cell carcinoma.
- AKA mucinous tubular and spindle cell carcinoma of the kidney.[75]
General
Microscopic
Features:[77]
- Mucin - may be scant.
- Spindle cells.
DDx:
- Sarcomatoid papillary renal cell carcinoma.[78]
Images:
IHC
Features:[79]
- CD10 -ve.
- AE1/AE3 +ve.
- AMACR +ve.
- CK7 +ve.
Collecting duct carcinoma
Renal medullary carcinoma
General
- Rare.
- Usually young adults.
- Associated with sickle cell trait (heterozygotes for the sickle cell allele).[80]
- Aggressive/poor prognosis.[81]
Aside:
- Kidney disease assoc. with sickle cell disorders:[80]
- Papillary necrosis.
- Nephrotic syndrome.
- Renal infarction.
- Pyelonephritis.
Gross
Features:[81]
- Well circumscribed.
- Renal medulla.
Microscopic
- Variable architecture:
- Reticular - classic.
- Adenoid cystic carcinoma-like appearance:
- Cystic spaces.
- Yolk sac-like.
- Tubular.
- Desmoplastic stroma - prominent.
- Inflammation:
- Lymphocytes.
- Neutrophils - margination in vessels.
DDx:
Image:
IHC
- SMARCB1 (INI1) -ve.[82]
Tubulocystic carcinoma of the kidney
Acquired cystic disease-associated renal cell carcinoma
Pediatric
The most common is nephroblastoma (Wilms tumour).
Others include:
- Metanephric stromal tumour.
- Metanephric adenofibroma
- Metanephric adenoma.
- Clear cell sarcoma of the kidney.
See also
- Urinary bladder.
- Medical kidney.
- Malakoplakia - yellow lesion on gross; may mimic RCC.
References
- ↑ Humphrey, Peter A; Dehner, Louis P; Pfeifer, John D (2008). The Washington Manual of Surgical Pathology (1st ed.). Lippincott Williams & Wilkins. pp. 288. ISBN 978-0781765275.
- ↑ Zhou, Ming; Magi-Galluzzi, Cristina (2006). Genitourinary Pathology: A Volume in Foundations in Diagnostic Pathology Series (1st ed.). Churchill Livingstone. pp. 281-304. ISBN 978-0443066771.
- ↑ 3.0 3.1 Srigley, JR.; Delahunt, B. (Jun 2009). "Uncommon and recently described renal carcinomas.". Mod Pathol 22 Suppl 2: S2-S23. doi:10.1038/modpathol.2009.70. PMID 19494850.
- ↑ Tickoo, SK.; Amin, MB. (Dec 1998). "Discriminant nuclear features of renal oncocytoma and chromophobe renal cell carcinoma. Analysis of their potential utility in the differential diagnosis.". Am J Clin Pathol 110 (6): 782-7. PMID 9844591.
- ↑ Humphrey, Peter A; Dehner, Louis P; Pfeifer, John D (2008). The Washington Manual of Surgical Pathology (1st ed.). Lippincott Williams & Wilkins. pp. 291. ISBN 978-0781765275.
- ↑ URL:http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0001544/. Accessed on: 14 July 2011.
- ↑ 7.0 7.1 7.2 Humphrey, Peter A; Dehner, Louis P; Pfeifer, John D (2008). The Washington Manual of Surgical Pathology (1st ed.). Lippincott Williams & Wilkins. pp. 289. ISBN 978-0781765275.
- ↑ Schmid HP, Szabo J (May 1997). "[Renal cell carcinoma--a current review]" (in German). Praxis (Bern 1994) 86 (20): 837?3. PMID 9312811.
- ↑ 9.0 9.1 Cotran, Ramzi S.; Kumar, Vinay; Fausto, Nelson; Nelso Fausto; Robbins, Stanley L.; Abbas, Abul K. (2005). Robbins and Cotran pathologic basis of disease (7th ed.). St. Louis, Mo: Elsevier Saunders. pp. 1016. ISBN 0-7216-0187-1.
- ↑ Capitanio, U.; Cloutier, V.; Zini, L.; Isbarn, H.; Jeldres, C.; Shariat, SF.; Perrotte, P.; Antebi, E. et al. (Jun 2009). "A critical assessment of the prognostic value of clear cell, papillary and chromophobe histological subtypes in renal cell carcinoma: a population-based study.". BJU Int 103 (11): 1496-500. doi:10.1111/j.1464-410X.2008.08259.x. PMID 19076149.
- ↑ Delahunt, B.; Bethwaite, PB.; Nacey, JN. (Oct 2007). "Outcome prediction for renal cell carcinoma: evaluation of prognostic factors for tumours divided according to histological subtype.". Pathology 39 (5): 459-65. doi:10.1080/00313020701570061. PMID 17886093.
- ↑ Zhou M, Roma A, Magi-Galluzzi C (June 2005). "The usefulness of immunohistochemical markers in the differential diagnosis of renal neoplasms". Clin. Lab. Med. 25 (2): 247?7. doi:10.1016/j.cll.2005.01.004. PMID 15848735.
- ↑ 13.0 13.1 Zhou M, Roma A, Magi-Galluzzi C (June 2005). "The usefulness of immunohistochemical markers in the differential diagnosis of renal neoplasms". Clin. Lab. Med. 25 (2): 247?7. doi:10.1016/j.cll.2005.01.004. PMID 15848735.
- ↑ 14.0 14.1 Langner C, Ratschek M, Rehak P, Schips L, Zigeuner R (February 2004). "Expression of MUC1 (EMA) and E-cadherin in renal cell carcinoma: a systematic immunohistochemical analysis of 188 cases". Mod. Pathol. 17 (2): 180?. doi:10.1038/modpathol.3800032. PMID 14657952.
- ↑ Liu L, Qian J, Singh H, Meiers I, Zhou X, Bostwick DG (August 2007). "Immunohistochemical analysis of chromophobe renal cell carcinoma, renal oncocytoma, and clear cell carcinoma: an optimal and practical panel for differential diagnosis". Arch. Pathol. Lab. Med. 131 (8): 1290?. PMID 17683191. http://journals.allenpress.com/jrnlserv/?request=get-abstract&issn=0003-9985&volume=131&page=1290.
- ↑ Pan CC, Chen PC, Ho DM (November 2004). "The diagnostic utility of MOC31, BerEP4, RCC marker and CD10 in the classification of renal cell carcinoma and renal oncocytoma: an immunohistochemical analysis of 328 cases". Histopathology 45 (5): 452?. doi:10.1111/j.1365-2559.2004.01962.x. PMID 15500648.
- ↑ 17.0 17.1 Geramizadeh B, Ravanshad M, Rahsaz M (2008). "Useful markers for differential diagnosis of oncocytoma, chromophobe renal cell carcinoma and conventional renal cell carcinoma". Indian J Pathol Microbiol 51 (2): 167?1. PMID 18603673. http://www.ijpmonline.org/article.asp?issn=0377-4929;year=2008;volume=51;issue=2;spage=167;epage=171;aulast=Geramizadeh.
- ↑ 18.0 18.1 Memeo L, Jhang J, Assaad AM, et al. (February 2007). "Immunohistochemical analysis for cytokeratin 7, KIT, and PAX2: value in the differential diagnosis of chromophobe cell carcinoma". Am. J. Clin. Pathol. 127 (2): 225–9. doi:10.1309/9KWEA4W9Y94D1AEE. PMID 17210525. http://ajcp.ascpjournals.org/cgi/pmidlookup?view=long&pmid=17210525.
- ↑ Mazal PR, Exner M, Haitel A, et al. (January 2005). "Expression of kidney-specific cadherin distinguishes chromophobe renal cell carcinoma from renal oncocytoma". Hum. Pathol. 36 (1): 22–8. doi:10.1016/j.humpath.2004.09.011. PMID 15712178.
- ↑ Krueger S, Sotlar K, Kausch I, Horny HP (2005). "Expression of KIT (CD117) in renal cell carcinoma and renal oncocytoma". Oncology 68 (2-3): 269-75. doi:10.1159/000086783. PMID 16015044.
- ↑ Martignoni G, Brunelli M, Gobbo S, et al (February 2007). "Role of molecular markers in diagnosis and prognosis of renal cell carcinoma". Anal. Quant. Cytol. Histol. 29 (1): 41?. PMID 17375873.
- ↑ Avery AK, Beckstead J, Renshaw AA, Corless CL (February 2000). "Use of antibodies to RCC and CD10 in the differential diagnosis of renal neoplasms". Am. J. Surg. Pathol. 24 (2): 203?0. PMID 10680888. http://meta.wkhealth.com/pt/pt-core/template-journal/lwwgateway/media/landingpage.htm?issn=0147-5185&volume=24&issue=2&spage=203.
- ↑ Kim MK, Kim S (December 2002). "Immunohistochemical profile of common epithelial neoplasms arising in the kidney". Appl. Immunohistochem. Mol. Morphol. 10 (4): 332–8. PMID 12613443.
- ↑ 24.0 24.1 de Peralta-Venturina, M.; Moch, H.; Amin, M.; Tamboli, P.; Hailemariam, S.; Mihatsch, M.; Javidan, J.; Stricker, H. et al. (Mar 2001). "Sarcomatoid differentiation in renal cell carcinoma: a study of 101 cases.". Am J Surg Pathol 25 (3): 275-84. PMID 11224597.
- ↑ 25.0 25.1 Lopez-Beltran, A.; Scarpelli, M.; Montironi, R.; Kirkali, Z. (May 2006). "2004 WHO classification of the renal tumors of the adults.". Eur Urol 49 (5): 798-805. doi:10.1016/j.eururo.2005.11.035. PMID 16442207.
- ↑ Cangiano, T.; Liao, J.; Naitoh, J.; Dorey, F.; Figlin, R.; Belldegrun, A. (Feb 1999). "Sarcomatoid renal cell carcinoma: biologic behavior, prognosis, and response to combined surgical resection and immunotherapy.". J Clin Oncol 17 (2): 523-8. PMID 10080595.
- ↑ 27.0 27.1 27.2 27.3 Dall'Oglio, MF.; Lieberknecht, M.; Gouveia, V.; Sant'Anna, AC.; Leite, KR.; Srougi, M.. "Sarcomatoid differentiation in renal cell carcinoma: prognostic implications.". Int Braz J Urol 31 (1): 10-6. PMID 15763002.
- ↑ Golshayan, AR.; George, S.; Heng, DY.; Elson, P.; Wood, LS.; Mekhail, TM.; Garcia, JA.; Aydin, H. et al. (Jan 2009). "Metastatic sarcomatoid renal cell carcinoma treated with vascular endothelial growth factor-targeted therapy.". J Clin Oncol 27 (2): 235-41. doi:10.1200/JCO.2008.18.0000. PMID 19064974.
- ↑ 29.0 29.1 29.2 Humphrey, Peter A; Dehner, Louis P; Pfeifer, John D (2008). The Washington Manual of Surgical Pathology (1st ed.). Lippincott Williams & Wilkins. pp. 290. ISBN 978-0781765275.
- ↑ Online 'Mendelian Inheritance in Man' (OMIM) 136850
- ↑ Online 'Mendelian Inheritance in Man' (OMIM) 135150
- ↑ Online 'Mendelian Inheritance in Man' (OMIM) 314310
- ↑ Verine, J.; Pluvinage, A.; Bousquet, G.; Lehmann-Che, J.; de Bazelaire, C.; Soufir, N.; Mongiat-Artus, P. (Nov 2010). "Hereditary renal cancer syndromes: an update of a systematic review.". Eur Urol 58 (5): 701-10. doi:10.1016/j.eururo.2010.08.031. PMID 20817385.
- ↑ Online 'Mendelian Inheritance in Man' (OMIM) 164860
- ↑ Zhou, Ming; Magi-Galluzzi, Cristina (2006). Genitourinary Pathology: A Volume in Foundations in Diagnostic Pathology Series (1st ed.). Churchill Livingstone. pp. 282. ISBN 978-0443066771.
- ↑ Delahunt, B.; Sika-Paotonu, D.; Bethwaite, PB.; McCredie, MR.; Martignoni, G.; Eble, JN.; Jordan, TW. (Jun 2007). "Fuhrman grading is not appropriate for chromophobe renal cell carcinoma.". Am J Surg Pathol 31 (6): 957-60. doi:10.1097/01.pas.0000249446.28713.53. PMID 17527087.
- ↑ AE. June 2010.
- ↑ 38.0 38.1 38.2 Humphrey, Peter A; Dehner, Louis P; Pfeifer, John D (2008). The Washington Manual of Surgical Pathology (1st ed.). Lippincott Williams & Wilkins. pp. 292. ISBN 978-0781765275.
- ↑ Agarwal, S.; Agrawal, U.; Mohanty, NK.; Saxena, S. (Mar 2011). "Multilocular cystic renal cell carcinoma: a case report of a rare entity.". Arch Pathol Lab Med 135 (3): 290-2. doi:10.1043/2010-0243-LE.1. PMID 21366448.
- ↑ You, D.; Shim, M.; Jeong, IG.; Song, C.; Kim, JK.; Ro, JY.; Hong, JH.; Ahn, H. et al. (Jul 2011). "Multilocular cystic renal cell carcinoma: clinicopathological features and preoperative prediction using multiphase computed tomography.". BJU Int. doi:10.1111/j.1464-410X.2011.10247.x. PMID 21722289.
- ↑ 41.0 41.1 Karakiewicz, PI.; Hutterer, GC.; Trinh, QD.; Pantuck, AJ.; Klatte, T.; Lam, JS.; Guille, F.; de La Taille, A. et al. (Oct 2007). "Unclassified renal cell carcinoma: an analysis of 85 cases.". BJU Int 100 (4): 802-8. doi:10.1111/j.1464-410X.2007.07148.x. PMID 17822461.
- ↑ Humphrey, Peter A; Dehner, Louis P; Pfeifer, John D (2008). The Washington Manual of Surgical Pathology (1st ed.). Lippincott Williams & Wilkins. pp. 293. ISBN 978-0781765275.
- ↑ 43.0 43.1 Lopez-Beltran, A.; Kirkali, Z.; Montironi, R.; Blanca, A.; Algaba, F.; Scarpelli, M.; Yorukoglu, K.; Hartmann, A. et al. (Sep 2012). "Unclassified renal cell carcinoma: a report of 56 cases.". BJU Int 110 (6): 786-93. doi:10.1111/j.1464-410X.2012.10934.x. PMID 22404824.
- ↑ Ishihara, A.; Yamashita, Y.; Takamori, H.; Kuroda, N. (Sep 2011). "Renal carcinoma with (6;11)(p21;q12) translocation: Report of an adult case.". Pathol Int 61 (9): 539-45. doi:10.1111/j.1440-1827.2011.02711.x. PMID 21884304.
- ↑ Humphrey, Peter A; Dehner, Louis P; Pfeifer, John D (2008). The Washington Manual of Surgical Pathology (1st ed.). Lippincott Williams & Wilkins. pp. 281. ISBN 978-0781765275.
- ↑ 46.0 46.1 Wang, KL.; Weinrach, DM.; Luan, C.; Han, M.; Lin, F.; Teh, BT.; Yang, XJ. (Feb 2007). "Renal papillary adenoma--a putative precursor of papillary renal cell carcinoma.". Hum Pathol 38 (2): 239-46. doi:10.1016/j.humpath.2006.07.016. PMID 17056094.
- ↑ Zhou, Ming; Magi-Galluzzi, Cristina (2006). Genitourinary Pathology: A Volume in Foundations in Diagnostic Pathology Series (1st ed.). Churchill Livingstone. pp. 288. ISBN 978-0443066771.
- ↑ 48.0 48.1 Algaba, F. (2008). "Renal adenomas: pathological differential diagnosis with malignant tumors.". Adv Urol: 974848. doi:10.1155/2008/974848. PMC 2563151. PMID 18846240. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2563151/.
- ↑ Seyam, RM.; Bissada, NK.; Kattan, SA.; Mokhtar, AA.; Aslam, M.; Fahmy, WE.; Mourad, WA.; Binmahfouz, AA. et al. (Nov 2008). "Changing trends in presentation, diagnosis and management of renal angiomyolipoma: comparison of sporadic and tuberous sclerosis complex-associated forms.". Urology 72 (5): 1077-82. doi:10.1016/j.urology.2008.07.049. PMID 18805573.
- ↑ Abrams, J.; Yee, DC.; Clark, TW. (Jul 2011). "Transradial embolization of a bleeding renal angiomyolipoma.". Vasc Endovascular Surg 45 (5): 470-3. doi:10.1177/1538574411408352. PMID 21571778.
- ↑ Zhang, SH.; Cong, WM.; Xian, ZH.; Wu, WQ.; Dong, H.; Wu, MC. (Oct 2004). "[Morphologic variants and immunohistochemical features of hepatic angiomyolipoma.]". Zhonghua Bing Li Xue Za Zhi 33 (5): 437-40. PMID 15498214.
- ↑ Nelson, CP.; Sanda, MG. (Oct 2002). "Contemporary diagnosis and management of renal angiomyolipoma.". J Urol 168 (4 Pt 1): 1315-25. doi:10.1097/01.ju.0000028200.86216.b2. PMID 12352384.
- ↑ Aydin, H.; Magi-Galluzzi, C.; Lane, BR.; Sercia, L.; Lopez, JI.; Rini, BI.; Zhou, M. (Feb 2009). "Renal angiomyolipoma: clinicopathologic study of 194 cases with emphasis on the epithelioid histology and tuberous sclerosis association.". Am J Surg Pathol 33 (2): 289-97. doi:10.1097/PAS.0b013e31817ed7a6. PMID 18852677.
- ↑ 54.0 54.1 Crapanzano, JP. (Jan 2005). "Fine-needle aspiration of renal angiomyolipoma: cytological findings and diagnostic pitfalls in a series of five cases.". Diagn Cytopathol 32 (1): 53-7. doi:10.1002/dc.20179. PMID 15584043.
- ↑ Aljerian, K.; Evans, AJ. (Oct 2004). "Pathologic quiz case: a 44-year-old woman with an incidental asymptomatic renal mass. Atypical epithelioid angiomyolipoma.". Arch Pathol Lab Med 128 (10): 1176-8. doi:10.1043/1543-2165(2004)1281176:PQCAYW2.0.CO;2. PMID 15387699.
- ↑ 56.0 56.1 Zhou, Ming; Magi-Galluzzi, Cristina (2006). Genitourinary Pathology: A Volume in Foundations in Diagnostic Pathology Series (1st ed.). Churchill Livingstone. pp. 324. ISBN 978-0443066771.
- ↑ Ooi, SM.; Vivian, JB.; Cohen, RJ. (2009). "The use of the Ki-67 marker in the pathological diagnosis of the epithelioid variant of renal angiomyolipoma.". Int Urol Nephrol 41 (3): 559-65. doi:10.1007/s11255-008-9473-1. PMID 18839327.
- ↑ 58.0 58.1 Afgan F, Mumtaz S, Ather MH (2007). "Preoperative diagnosis of xanthogranulomatous pyelonephritis". Urol J 4 (3): 169–73. PMID 17987581.
- ↑ 59.0 59.1 59.2 Al-Ghazo MA, Ghalayini IF, Matalka II, Al-Kaisi NS, Khader YS (October 2006). "Xanthogranulomatous pyelonephritis: Analysis of 18 cases". Asian J Surg 29 (4): 257–61. PMID 17098659.
- ↑ 60.0 60.1 Wong, L.; Hsu, TH.; Perlroth, MG.; Hofmann, LV.; Haynes, CM.; Katznelson, L. (Feb 2008). "Reninoma: case report and literature review.". J Hypertens 26 (2): 368-73. doi:10.1097/HJH.0b013e3282f283f3. PMID 18192852.
- ↑ 61.0 61.1 61.2 Chao, CT.; Chang, FC.; Wu, VC.; Chen, JC. (Jan 2011). "Reninoma.". Kidney Int 79 (2): 260. doi:10.1038/ki.2010.445. PMID 21191395.
- ↑ 62.0 62.1 Hanna, W.; Tepperman, B.; Logan, AG.; Robinette, MA.; Colapinto, R.; Phillips, MJ. (Apr 1979). "Juxtaglomerular cell tumour (reninoma) with paroxysmal hypertension.". Can Med Assoc J 120 (8): 957-9. PMID PMC = 1819229 436071 PMC = 1819229.
- ↑ URL: http://path.upmc.edu/cases/case29/micro.html. Accessed on: 18 December 2011.
- ↑ Bircan, S.; Orhan, D.; Tulunay, O.; Safak, M. (2000). "Renomedullary interstitial cell tumor.". Urol Int 65 (3): 163-6. PMID 11054036.
- ↑ 65.0 65.1 Humphrey, Peter A; Dehner, Louis P; Pfeifer, John D (2008). The Washington Manual of Surgical Pathology (1st ed.). Lippincott Williams & Wilkins. pp. 295. ISBN 978-0781765275.
- ↑ 66.0 66.1 Tsurukawa, H.; Iuchi, H.; Osanai, H.; Yamaguchi, S.; Hashimoto, H.; Kaneko, S.; Yachiku, S. (Jan 2000). "[Renomedullary interstitial cell tumor: a case report].". Nihon Hinyokika Gakkai Zasshi 91 (1): 37-40. PMID 10689882.
- ↑ Kozłowska, J.; Okoń, K. (2008). "Renal tumors in postmortem material.". Pol J Pathol 59 (1): 21-5. PMID 18655367.
- ↑ URL: http://webpathology.com/image.asp?n=16&Case=71. Accessed on: 17 October 2011.
- ↑ Kuroda, N.; Toi, M.; Miyazaki, E.; Hayashi, Y.; Nakayama, H.; Hiroi, M.; Enzan, H.. "Participation of alpha-smooth muscle actin-positive cells in renomedullary interstitial cell tumors.". Oncol Rep 9 (4): 745-50. PMID 12066202.
- ↑ Le Nué, R.; Marcellin, L.; Ripepi, M.; Henry, C.; Kretz, JM.; Geiss, S. (Aug 2011). "Conservative treatment of metanephric adenoma. A case report and review of the literature.". J Pediatr Urol 7 (4): 399-403. doi:10.1016/j.jpurol.2010.09.010. PMID 21220212.
- ↑ 71.0 71.1 Humphrey, Peter A; Dehner, Louis P; Pfeifer, John D (2008). The Washington Manual of Surgical Pathology (1st ed.). Lippincott Williams & Wilkins. pp. 284. ISBN 978-0781765275.
- ↑ 72.0 72.1 72.2 Watanabe, S.; Naganuma, H.; Shimizu, M.; Ota, S.; Murata, S.; Nihei, N.; Matsushima, J.; Mikami, S. et al. (2013). "Adult nephroblastoma with predominant epithelial component: a differential diagnostic candidate of papillary renal cell carcinoma and metanephric adenoma-report of three cases.". Case Rep Pathol 2013: 675875. doi:10.1155/2013/675875. PMID 24083046.
- ↑ 73.0 73.1 73.2 Turbiner, J.; Amin, MB.; Humphrey, PA.; Srigley, JR.; De Leval, L.; Radhakrishnan, A.; Oliva, E. (Apr 2007). "Cystic nephroma and mixed epithelial and stromal tumor of kidney: a detailed clinicopathologic analysis of 34 cases and proposal for renal epithelial and stromal tumor (REST) as a unifying term.". Am J Surg Pathol 31 (4): 489-500. doi:10.1097/PAS.0b013e31802bdd56. PMID 17414095.
- ↑ 74.0 74.1 Mai, KT.; Elkeilani, A.; Veinot, JP. (Apr 2007). "Mixed epithelial and stromal tumour (MEST) of the kidney: report of 14 cases with male and PEComatous variants and proposed histopathogenesis.". Pathology 39 (2): 235-40. doi:10.1080/00313020701230799. PMID 17454754. Cite error: Invalid
<ref>tag; name "pmid17454754" defined multiple times with different content - ↑ Brandal, P.; Lie, AK.; Bassarova, A.; Svindland, A.; Risberg, B.; Danielsen, H.; Heim, S. (Feb 2006). "Genomic aberrations in mucinous tubular and spindle cell renal cell carcinomas.". Mod Pathol 19 (2): 186-94. doi:10.1038/modpathol.3800499. PMID 16258504.
- ↑ Geramizadeh B, Salehipour M, Moradi A (2009). "Mucinous tubular and spindle cell carcinoma of kidney: a rare case report and review of the literature". Indian J Pathol Microbiol 52 (4): 514–6. doi:10.4103/0377-4929.56144. PMID 19805959.
- ↑ Trabelsi A, Stita W, Yacoubi MT, Rammeh S, Hmissa S, Korbi S (December 2008). "Renal mucinous tubular and spindle cell carcinoma". Can Urol Assoc J 2 (6): 635–6. PMC 2593603. PMID 19066686. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2593603/.
- ↑ Dhillon J, Amin MB, Selbs E, Turi GK, Paner GP, Reuter VE (January 2009). "Mucinous tubular and spindle cell carcinoma of the kidney with sarcomatoid change". Am. J. Surg. Pathol. 33 (1): 44–9. doi:10.1097/PAS.0b013e3181829ed5. PMID 18941398.
- ↑ Ferlicot S, Allory Y, Compérat E, et al. (December 2005). "Mucinous tubular and spindle cell carcinoma: a report of 15 cases and a review of the literature". Virchows Arch. 447 (6): 978–83. doi:10.1007/s00428-005-0036-x. PMID 16231179.
- ↑ 80.0 80.1 80.2 Davis CJ, Mostofi FK, Sesterhenn IA (January 1995). "Renal medullary carcinoma. The seventh sickle cell nephropathy". Am. J. Surg. Pathol. 19 (1): 1–11. PMID 7528470.
- ↑ 81.0 81.1 81.2 Watanabe, IC.; Billis, A.; Guimarães, MS.; Alvarenga, M.; de Matos, AC.; Cardinalli, IA.; Filippi, RZ.; de Castro, MG. et al. (Sep 2007). "Renal medullary carcinoma: report of seven cases from Brazil.". Mod Pathol 20 (9): 914-20. doi:10.1038/modpathol.3800934. PMID 17643096. http://www.nature.com/modpathol/journal/v20/n9/full/3800934a.html. Cite error: Invalid
<ref>tag; name "pmid17643096" defined multiple times with different content Cite error: Invalid<ref>tag; name "pmid17643096" defined multiple times with different content - ↑ Humphrey, Peter A; Dehner, Louis P; Pfeifer, John D (2008). The Washington Manual of Surgical Pathology (1st ed.). Lippincott Williams & Wilkins. pp. 286. ISBN 978-0781765275.