Invasive breast cancer
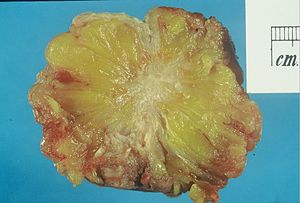
The article deals with invasive breast cancer and the evaluation of hormone receptor & HER2 status. Non-invasive breast cancer is dealt with in non-invasive breast cancer.
Introduction
Overview of invasive breast cancer subtypes
Common epithelial subtypes
Type and percentage of breast carcinomas:[1]
- Ductal - AKA no special type (NST) - 79%.
- Lobular - 10%.
- Cribriform / tubular - 6%.
- Mucinous (colloid) - 2%.
- Medullary - 2%.
- Papillary - 1%.
- Metaplastic - <1%.
Common stromal types
- Malignant phyllodes tumour.
- Angiosarcoma - post-radiation ~ 10 years.[2]
Good prognosis subtypes
Three good prognosis subtypes:[3]
- Tubular carcinoma.
- Mucinous carcinoma.
- Papillary carcinoma.
Comprehensive list of invasive breast cancer subtypes
Epithelial
Counterparts of in situ lesions:
- Invasive ductal carinoma, not otherwise specified.
- Invasive lobular carcinoma.
- Invasive cribriform carcinoma.
- Invasive papillary carcinoma.
- Invasive micropapillary carcinoma.
Other epithelial tumours:
- Tubular carcinoma.
- Medullary carcinoma.
- Mucinous carinoma.
- Metaplastic carcinoma.
- Neuroendocrine tumour.
- Apocrine carcinoma.
- Lipid-rich carcinoma.
- Secretory carcinoma.
- Oncocytic carcinoma.
- Glycogen-rich clear cell carcinoma.
Epithelial tumours seen in the salivary gland:
Seen in the skin:
Clinically diagnosed:
- Inflammatory carcinoma.
In situ lesions:
Proliferative lesions:
Non-specific:
- Microinvasive carcinoma.
Papillary:
- Papilloma.
- Atypical papilloma.
- Intraductal papillary carcinoma.
Adenomas:
- Ductal adenoma.
- Tubular adenoma.
- Lactating adenoma.
- Apocrine adenoma.
- Pleomorphic adenoma.
Myoepithelial
- Myoepitheliosis.
- Adenomyoepithelial adenosis.
- Adenomyoepithelioma.
- Malignant adenomyoepithelioma.
Mesenchymal tumours
- See: Soft tissue lesions.
Fibroepithelial tumours
- Fibroadenoma.
- Phyllodes tumour.
- Periductal stromal sarcoma, low grade.
- Mammary hamartoma.
Nipple lesions
- Nipple adenoma.
- Syringomatous adenoma.
- Paget disease of the breast.
Other
Familial breast cancer
Breast IHC
Molecular classification of invasive carcinoma
A molecular classification:[4]
| Type | Percentage | IHC | Histology | Prognosis/clinical |
|---|---|---|---|---|
| Luminal A | ~45% | ER+ PR+ HER2- | well-differentiated | good, chemo resistant |
| Luminal B | 17% | ER+ PR+ HER2+ | high grade | poor, +/- chemo responsive |
| Normal breast-like | ~8% | ER+ PR+ (?) HER2- | well-differentiated | good |
| Basal-like | ~20% | ER- PR- HER2- | poorly differentiated | aggressive, may have good chemo response, classic for BRCA1 mutation |
| HER2 positive | ~10% | ER- PR- (?) HER2+ | poorly differentiated | poor |
The above is not applied clinically. A panel of immunostains (ER, PR, HER2, EGFR, CK5/6) can reproduce the molecular groupings; however, these groupings originate from gene expression profiling studies[5]
A newer classification outlines 10 subtypes based on molecular drivers identified by analysis of genomic and transcriptomic data from 2,000 breast tumors.[6]
Basal-like breast carcinoma
- Overview:[7]
- A category of breast carcinomas defined by gene expression profiling.
- Not used in clinical practice.
- Somewhere between 15-30% of breast carcinomas.
- Can be roughly be identified by immunohistochemistry - basal markers (CK14, p63, calponin, SMA).
- Not derived from myoepithelial cells, merely express a phenotype more in keeping with basal cells than ductal cells.
- Most triple negative (ER, PgR, Her-2); therefore cannot be treated with the usual therapeutic agents.
- There is an association in young women between basal-like breast cancer and BRCA1 mutation.
- Discussions of BRCA1 associated tumors, TNBC and BLBC are typically muddied by the overlap.
- Increased incidence in some populations - African-Americans, young women
- Sporadic basal-like cancers do not have a BRCA1 mutation but may have a dysfunctional BRCA1 pathway.
- p53 mutations are frequent.
- This molecular group includes a variety of morphologic phenotypes including:
- High grade invasive ductal carcinoma of no special type.
- Medullary-like carcinoma (a carcinoma with some but not all the features of medullary carcinoma).
- Medullary carcinoma
- Metaplastic carcinoma.
- Adenoid cystic carcinoma.
- Secretory carcinoma.
- Classic morphological clues of a basal type cancer usually refer to medullary carcinoma features:
- Relatively circumscribed.
- Geographic necrosis.
- Abundant mitoses.
- Pushing margins.
- Central fibrosis or necrosis.
- High histological grade.
- Exceptionally high mitotic rate.
- Pushing borders.
- Conspicuous lymphocytic infiltrate.
- Behaviour:
- Basal-like breast cancer is a heterogeneous group.
- The behaviour of basal-like breast cancer appears to fall into two groups:
- The tumours that are by nature low grade (ie adenoid cystic carcinoma) and/or do not metastasise have a better prognosis than other types of breast carcinoma.
- The tumours with early metastasis that may behave more aggressively
- Hematogenous spread -greater tendency to metastasise to visceral sites (notably lung and brain) instead of to nodes and bone.
- Many have a complete response to chemotherapy and survival rates similar to typical breast cancer
- Non-complete response to chemotherapy is associated with low survival at 5 years.
Other sources Minireview: Basal-Like Breast Cancer: From Molecular Profiles to Targeted Therapies <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3035993/>
Triple Negative Breast Carcinoma
Features:[8]
- A category of breast carcinomas defined by immunohistochemical/FISH expression of ER, PR and HER2.
- Important to identify in clinical practice.
- About 15% of breast carcinomas.
- Important group due to a lack of tailored therapies for this group
- Some triple negatives also express androgen receptor and have and [apocrine carcinoma] morphology.[9]
- May respond to therapies targeting the androgen receptor.
- BCL11A overexpression recently identified as an oncogenic driver for some triple negatives [10]
- Targeted therapies may include inhibitors of BCL11A.
- Some triple negatives also express androgen receptor and have and [apocrine carcinoma] morphology.[9]
- Triple-negative and basal-like phenotypes are not synonymous but overlap
- About 70% of triple-negative tumours are basal-like.
- About 70% of basal-like tumors are triple-negative tumours.
- Discussions of BRCA1 associated tumors, TNBC and BLBC are typically muddied by the overlap.
- Classic 'morphological clues' to a triple negative cancer usually refer to medullary carcinoma features.
Immunostains for typing and diagnosis
DCIS versus LCIS
Tabular comparison for DCIS versus LCIS:[11][12]
| Disease | E-cadherin | Beta-catenin | 34betaE12 | CAM5.2 (CK8) |
|---|---|---|---|---|
| DCIS | +ve | +ve | -ve | +ve peripheral cytoplasm |
| LCIS | -ve | -ve | +ve perinuclear | +ve perinuclear |
Invasive versus non-invasive
Myoepithelial markers - typically lost in invasive carcinoma:[13]
| Stain | Location | Notes |
|---|---|---|
| p63 | nuclear | up to 10% of invasive tumours +ve[14] |
| Smooth muscle actin (SMA) | cytoplasmic | stains myofibroblasts & blood vessels |
| Calponin | cytoplasmic | stains myofibroblasts & blood vessels |
| Smooth muscle myosin heavy chain (SMM-HC) |
cytoplasmic | stains myofibroblasts & blood vessels |
Respecting findings that might indicate a more extensive search for microinvasion be undertaken in cases of pure ductal carcinoma in situ (DCIS), a recent study found 1) intermediate or high DCIS grade, 2) tumor thickness, and 3) diffuse peritumoral retraction clefts, but not such things as lymph node metastases, or HER2 score, independently increased the likelihood of finding a microinvasive component. [15]
Usual ductal hyperplasia versus ductal carcinoma in situ
Markers for UDH versus DCIS:[14]
| Disease | CK5/6 | ER |
|---|---|---|
| UDH | diffuse +ve | patchy +ve |
| DCIS | -ve | diffuse +ve |
Lymphovascular invasion
- D2-40 - marks the lymphatic spaces.[16][17]
- CD31 - marks lymphovascular spaces.
- CD34 - marks lymphovascular spaces, less specific than CD31.
Lymph node metastases
Immunostaining of sentinel lymph nodes to look for isolated tumour cells and small lymph node metastases may be done.
- CAM5.2 may be used.
- Not done routinely.
- ER (estrogen receptor).
- Positive in most breast cancers; +ve in ~75-80%.[18]
- PR (progesterone receptor).
- Positive in most breast cancers; +ve in ~65-70%.[18]
- HER2/neu (HER2).
Notes:
- Male breast cancer is usually hormone receptor positive (~97%), and HER2 positivity is quite rare (~6%).[21]
- ASCO/CAP guidelines recommend that cold ischemia time be <1 hour.[22]
ER & PR scoring
Nuclear staining:[18]
- Give a percentage, i.e. 0-100%.
- Important cut points: 1% and 10%.
- 0% = negative - not treated.
- <10% = low positivity - treated.
- Important cut points: 1% and 10%.
Notes:
- Normal breast epithelial cells have a patchy staining for ER and PR.
- Evaluated on the invasive component.
HER2 scoring
Immunohistochemical based testing:[23][24]
| Score | Staining intensity | Cells stained (%) | Membrane staining | Management | Percentage of cases |
|---|---|---|---|---|---|
| 0 | no staining/barely visible | ≤10% | incomplete | No HER2 blocker | ~60% |
| 1+ | minimal/barely visible | >10% | incomplete | No HER2 blocker | ~10% |
| 2+ | weak-to-moderate | >10% | incomplete (circumferential) | Needs SISH or FISH | ~10% † |
| 2+ | intense | ≤10% | complete | Needs SISH or FISH | ~10% † |
| 3+ | intense staining | >10% ‡ | complete | HER2 blocker | ~20% |
Note for IHC:
- Normal breast epithelial cells do not stain with HER2.
- Evaluated on the invasive component.
- SISH = silver in situ hybridization.
- FISH = fluorescence in situ hybridization.
- † Together approximately 10%.
- ‡ The cut point was 10%, changed to 30% and then changed back to 10%.[23]
ISH based testing:[25]
| Result | Ratio criteria | Gene copy number criteria |
|---|---|---|
| Positive | ≥2.0 HER2/CEP17 | ≥6.0 copies of HER2/cell |
| Equivocal | <2.0 HER2/CEP17 (required) | 4.0-6.0 copies of HER2/cell |
| Negative | <2.0 HER2/CEP17 | <4.0 copies of HER2/cell |
Note for ISH:
- Can be called positive based on either ratio criteria or gene copy number criteria.
Clinical
- ER & PR status determine whether a patient will get tamoxifen or other estrogen receptor modulators, such as raloxifene (Evista).
- HER2 status determines whether patient will get traztuzumab (Herceptin) or other HER2/neu modulators.
Characteristics of the subtypes
Invasive ductal carcinoma of the breast
Invasive lobular carcinoma
- Abbreviated ILC.
- AKA lobular carcinoma.
Medullary breast carcinoma
- AKA medullary carcinoma of the breast.
Tubular carcinoma of the breast
- AKA tubular carcinoma.
Metaplastic breast carcinoma
- AKA metaplastic carcinoma.
Invasive micropapillary carcinoma of the breast
- AKA micropapillary carcinoma.
Apocrine carcinoma of the breast
Mucinous breast carcinoma
Adenoid cystic carcinoma of the breast
- AKA breast adenoid cystic carcinoma.
Intracystic papillary breast carcinoma
- AKA encapsulated or encysted papillary carcinoma of the breast, abbreviated EPC.
Glycogen-rich clear cell carcinoma of the breast
- Abbreviated GRCC.
Secretory carcinoma of the breast
- AKA secretory breast carcinoma, abbreviated SBC.
Invasive cribriform carcinoma of the breast
Invasive papillary carcinoma of the breast
- Should not be confused with the indolent behaving intracystic papillary carcinoma of the breast, also known as encapsulated papillary carcinoma of the breast.
Grading breast cancer
Staging breast cancer
Lymphovascular invasion
In the context of breast pathology, the Rosen criteria for LVI are widely excepted, and are as follows:[26][27]
- Must be outside of the tumour proper.
- LVI is usually very close -- typically within 0.1 cm.
- Contour of cells should differ from possible vessel wall.
- DCIS with retraction artifact mimicing LVI has a contour that matches its surrounding fibrous tissue.
- Endothelium (usu. flat) should be visible.
- Lymphatics are found adjacent to blood vessels - vessels should be present in the vicinity.
Memory device LUBE-O:
- LVI has a Unique contour, Blood vessels and Endothelium in the vicinity, and is Outside of the tumour.
Note:
- LVI does not affect the stage.
Other
Paget's disease
General
- Associated with underlying breast carcinoma.[28]
Notes:
- Unrelated to Paget disease of the bone.
Microscopic
Features:[28]
- Cells in the epidermis:
- Epitheliod morphology (round/ovoid).
- Cells nested or single.
- Clear/pale cytoplasm key feature - may also be eosinophilic.
- Large nucleoli.
Images:
IHC & DDx:
- See Paget disease.
Trivia
Tumour size and lymph node metastases
There is a paper[29] that calculates the probability of lymph node mets based on tumour size. The developed formula is:
Where:
- = the probability of the lymph nodes being positive.
- D = the largest dimension of the tumour in millimetres.
- Z = 1.0041.
- = 0.019.
Selected values
| Tumour size (mm) | Probability |
| 5 | 9 % |
| 10 | 17 % |
| 15 | 25 % |
| 20 | 32 % |
| 25 | 38 % |
| 30 | 44 % |
| 35 | 49 % |
| 40 | 54 % |
| 45 | 58 % |
| 50 | 62 % |
Natural history
There is a theory that up to 22% of small (radiographically detected) breast tumours regress, based on an analysis in a large population.[30] The study is supported by NCI's SEER data.[31] Also, it generated many comments.[30]
Missed macrometastases
The effect of missed macrometastases is small; this implies using IHC to look for isolated tumour cells is money that isn't well spent.[32]
See also
References
- ↑ Cotran, Ramzi S.; Kumar, Vinay; Fausto, Nelson; Nelso Fausto; Robbins, Stanley L.; Abbas, Abul K. (2005). Robbins and Cotran pathologic basis of disease (7th ed.). St. Louis, Mo: Elsevier Saunders. pp. 1143. ISBN 0-7216-0187-1.
- ↑ URL: http://www.breastpathology.info/Case_of_the_month/2007/COTM_1007%20discussion.html. Accessed on: 28 November 2010.
- ↑ URL: http://emedicine.medscape.com/article/1947145-overview. Accessed on: 24 August 2012.
- ↑ Mitchell, Richard; Kumar, Vinay; Fausto, Nelson; Abbas, Abul K.; Aster, Jon (2011). Pocket Companion to Robbins & Cotran Pathologic Basis of Disease (8th ed.). Elsevier Saunders. pp. 547. ISBN 978-1416054542.
- ↑ Tang, P.; Skinner, KA.; Hicks, DG. (Sep 2009). "Molecular classification of breast carcinomas by immunohistochemical analysis: are we ready?". Diagn Mol Pathol 18 (3): 125-32. doi:10.1097/PDM.0b013e31818d107b. PMID 19704256.
- ↑ Curtis, C.; Shah, SP.; Chin, SF.; Turashvili, G.; Rueda, OM.; Dunning, MJ.; Speed, D.; Lynch, AG. et al. (Jun 2012). "The genomic and transcriptomic architecture of 2,000 breast tumours reveals novel subgroups.". Nature 486 (7403): 346-52. doi:10.1038/nature10983. PMID 22522925.
- ↑ Badve, S.; Dabbs, DJ.; Schnitt, SJ.; Baehner, FL.; Decker, T.; Eusebi, V.; Fox, SB.; Ichihara, S. et al. (Feb 2011). "Basal-like and triple-negative breast cancers: a critical review with an emphasis on the implications for pathologists and oncologists.". Mod Pathol 24 (2): 157-67. doi:10.1038/modpathol.2010.200. PMID 21076464.
- ↑ Badve, S.; Dabbs, DJ.; Schnitt, SJ.; Baehner, FL.; Decker, T.; Eusebi, V.; Fox, SB.; Ichihara, S. et al. (Feb 2011). "Basal-like and triple-negative breast cancers: a critical review with an emphasis on the implications for pathologists and oncologists.". Mod Pathol 24 (2): 157-67. doi:10.1038/modpathol.2010.200. PMID 21076464.
- ↑ Niemeier, LA.; Dabbs, DJ.; Beriwal, S.; Striebel, JM.; Bhargava, R. (Feb 2010). "Androgen receptor in breast cancer: expression in estrogen receptor-positive tumors and in estrogen receptor-negative tumors with apocrine differentiation.". Mod Pathol 23 (2): 205-12. doi:10.1038/modpathol.2009.159. PMID 19898421.
- ↑ Khaled, WT.; Choon Lee, S.; Stingl, J.; Chen, X.; Raza Ali, H.; Rueda, OM.; Hadi, F.; Wang, J. et al. (2015). "BCL11A is a triple-negative breast cancer gene with critical functions in stem and progenitor cells.". Nat Commun 6: 5987. doi:10.1038/ncomms6987. PMID 25574598.
- ↑ O'Malley, Frances P.; Pinder, Sarah E. (2006). Breast Pathology: A Volume in Foundations in Diagnostic Pathology series (1st ed.). Churchill Livingstone. pp. 275. ISBN 978-0443066801.
- ↑ Yeh IT, Mies C (March 2008). "Application of immunohistochemistry to breast lesions". Arch. Pathol. Lab. Med. 132 (3): 349-58. PMID 18318578. http://journals.allenpress.com/jrnlserv/?request=get-abstract&issn=0003-9985&volume=132&page=349.
- ↑ Lester, Susan Carole (2010). Manual of Surgical Pathology (3rd ed.). Saunders. pp. 88. ISBN 978-0-323-06516-0.
- ↑ 14.0 14.1 O'Malley, Frances P.; Pinder, Sarah E. (2006). Breast Pathology: A Volume in Foundations in Diagnostic Pathology series (1st ed.). Churchill Livingstone. pp. 276. ISBN 978-0443066801.
- ↑ Mori K, Takeda M, Kodama Y, Kiyokawa H, Yasojima H, Mizutani M, Otani Y, Morikawa N, Masuda N, Mano M (2017). "Tumor thickness and histological features as predictors of invasive foci within preoperatively diagnosed ductal carcinoma in situ". Human Pathology 64: 145-155. doi:10.1016/j.humpath.2017.04.004. PMID 28434924.
- ↑ Ordóñez NG (March 2006). "Podoplanin: a novel diagnostic immunohistochemical marker". Adv Anat Pathol 13 (2): 83-8. doi:10.1097/01.pap.0000213007.48479.94. PMID 16670463.
- ↑ Kahn HJ, Marks A (September 2002). "A new monoclonal antibody, D2-40, for detection of lymphatic invasion in primary tumors". Lab. Invest. 82 (9): 1255-7. PMID 12218087.
- ↑ 18.0 18.1 18.2 18.3 Lester, Susan Carole (2005). Manual of Surgical Pathology (2nd ed.). Saunders. pp. 241-2. ISBN 978-0443066450.
- ↑ Gallardo, A.; Lerma, E.; Escuin, D.; Tibau, A.; Muñoz, J.; Ojeda, B.; Barnadas, A.; Adrover, E. et al. (Apr 2012). "Increased signalling of EGFR and IGF1R, and deregulation of PTEN/PI3K/Akt pathway are related with trastuzumab resistance in HER2 breast carcinomas.". Br J Cancer 106 (8): 1367-73. doi:10.1038/bjc.2012.85. PMID 22454081.
- ↑ Jensen, JD.; Knoop, A.; Laenkholm, AV.; Grauslund, M.; Jensen, MB.; Santoni-Rugiu, E.; Andersson, M.; Ewertz, M. (Dec 2011). "PIK3CA mutations, PTEN, and pHER2 expression and impact on outcome in HER2-positive early-stage breast cancer patients treated with adjuvant chemotherapy and trastuzumab.". Ann Oncol. doi:10.1093/annonc/mdr546. PMID 22172323.
- ↑ Schildhaus, HU.; Schroeder, L.; Merkelbach-Bruse, S.; Binot, E.; Büttner, R.; Kuhn, W.; Rudlowski, C. (Sep 2013). "Therapeutic strategies in male breast cancer: Clinical implications of chromosome 17 gene alterations and molecular subtypes.". Breast. doi:10.1016/j.breast.2013.08.008. PMID 24080492.
- ↑ Yildiz-Aktas, IZ.; Dabbs, DJ.; Bhargava, R. (Aug 2012). "The effect of cold ischemic time on the immunohistochemical evaluation of estrogen receptor, progesterone receptor, and HER2 expression in invasive breast carcinoma.". Mod Pathol 25 (8): 1098-105. doi:10.1038/modpathol.2012.59. PMID 22460807.
- ↑ 23.0 23.1 Rakha, EA.; Starczynski, J.; Lee, AH.; Ellis, IO. (Apr 2014). "The updated ASCO/CAP guideline recommendations for HER2 testing in the management of invasive breast cancer: a critical review of their implications for routine practice.". Histopathology 64 (5): 609-15. doi:10.1111/his.12357. PMID 24382093.
- ↑ URL: http://www.cap.org/apps/docs/committees/cancer/cancer_protocols/breast_biomarker_template.pdf. Accessed on: October 7, 2014.
- ↑ URL: http://www.cap.org/apps/docs/committees/cancer/cancer_protocols/breast_biomarker_template.pdf. Accessed on: October 7, 2014.
- ↑ Rosen, PP. (1983). "Tumor emboli in intramammary lymphatics in breast carcinoma: pathologic criteria for diagnosis and clinical significance.". Pathol Annu 18 Pt 2: 215-32. PMID 6674861.
- ↑ URL: http://www.cap.org/apps/docs/committees/cancer/cancer_protocols/2009/InvasiveBreast_09protocol.pdf. Accessed on: 5 August 2011.
- ↑ 28.0 28.1 URL: http://emedicine.medscape.com/article/1101235-diagnosis
- ↑ Porembka, MR.; Abraham, RL.; Sefko, JA.; Deshpande, AD.; Jeffe, DB.; Margenthaler, JA. (Oct 2008). "Factors associated with lymph node assessment in ductal carcinoma in situ: analysis of 1988-2002 seer data.". Ann Surg Oncol 15 (10): 2709-19. doi:10.1245/s10434-008-9947-5. PMID 18483831. http://onlinelibrary.wiley.com/doi/10.1002/cncr.24592/pdf.
- ↑ 30.0 30.1 Zahl, PH.; Maehlen, J.; Welch, HG. (Nov 2008). "The natural history of invasive breast cancers detected by screening mammography.". Arch Intern Med 168 (21): 2311-6. doi:10.1001/archinte.168.21.2311. PMID 19029493.
- ↑ Jatoi, I.; Anderson, WF. (May 2009). "Breast cancer overdiagnosis with screening mammography.". Arch Intern Med 169 (10): 999-1000, author reply 1000-1. doi:10.1001/archinternmed.2009.95. PMID 19468099.
- ↑ Weaver, DL.; Ashikaga, T.; Krag, DN.; Skelly, JM.; Anderson, SJ.; Harlow, SP.; Julian, TB.; Mamounas, EP. et al. (Feb 2011). "Effect of occult metastases on survival in node-negative breast cancer.". N Engl J Med 364 (5): 412-21. doi:10.1056/NEJMoa1008108. PMID 21247310.


