Pulmonary cytopathology
Pulmonary cytopathology, also lung cytology, is a subset of cytopathology.
This article deals only with pulmonary cytopathology (FNAs, sputum samples). Pleural cavity specimens are dealt with in the mesothelial cytopathology article.
An introduction to cytopathology is in the cytopathology article. An introduction to lung pathology is in the lung article.
Introduction
Specimen types
- Bronchial brushings.
- Bronchial washing (contain airway cells).
- Bronchoalveolar lavage (BAL).
- Endobronchial ultrasongraphic transbronchial needle aspiration (EBUS-TNA).
- Endoscopic ultrasound-guided fine needle aspiration.
Adequacy criteria
EBUS
There is a proposed standard for EBUS specimens:[1]
- > 5 low power fields (×10 objective) with >= 100 lymphocytes and < 2 groups of bronchial cells.
Note:
- A simplified version: 5 fields of view x 100 cells/1 field of view = 500 cells.
Other specimens
Brushings, washes and lavages:
- One should see pulmonary macrophages (large cells with bubbly green/brown cytoplasm, eccentric reniform nucleus).
Notes:
- Ciliated cells may be from the nasopharynx - not proof of lung parenchymal tissue.
- There is no generally accepted standard for bronchial brushings, washes and lavages. A house standard at a larger teaching centre is:[2]
- Sputum: >= 10 pulmonary macrophages.
Normal cytology
- Cells with cilia = good.
- Cells with "terminal bar" (band at luminal aspect of cell - associated with cilia) = good.
Images
Cartilage
Epithelium
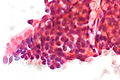
Reactive bronchial cells
Cytology
Features:
- Clusters of small round cells.
- +/-Grooves.
Images
BBE - very high mag.
RBCs - extremely high mag.
www
Actinomycetes
Image:
Infection
If you see lotsa lymphocytes think tumour.[5]
Pneumocystis pneumonia
- Abbreviated PCP.
Features:
- Casts of frothy material/large proteinaceous debris - approximately the size of an alveolus.
DDx:
Aspergillosis
Features:
- Hyphae... branching at 45 degrees.
Image:
Zygomycosis
- AKA mucormycosis.
Features:
- Hyphae... with variable width.
Image:
Crytococcus
Features:
- Prominent (i.e. thick polysaccharide) capsule.
- Seen well on Pap stain... harder to see on rapid Romanowsky stain.
- Spherical - 5-15 micrometres.
DDx:
- Bastomycosis.
- Doesn't have thick capsule
- Has broad based budding.
- Coccidioidomycosis - larger (20-60 micrometers).
Image:
Cancer
Approaches to lung cancer
Lung cancer in a table
| Small cell carcinoma | Adenocarcinoma | Squamous cell carcinoma | Value | |
|---|---|---|---|---|
| Cellular cohesion | Single cells/stripped nuclei common | Cohesive | Cohesive | Cohesive (only) suggests NSCLC |
| Nuclear moulding | Present | Absent | Absent | R/i & r/o: small cell carcinoma and carcinoid |
| Small nucleoli (difficult to see on Field stain) | Multiple pseudo-nucleoli may be seen in occ. cells | Many be present | Often present | Weak discriminative valuable |
| Large nucleoli | Never | Present | Rarely | R/i adenocarcinoma; should prompt consideration of melanoma briefly |
| Location of nucleus | Eccentric | Eccentric | Central | Useful for SCC vs. adenocarcinoma |
| Cytoplasm | Scant | Abundant, bubbly, +/-vacuole with mucin | Abundant, "dense" | Abundant r/o small cell, mucin vacuole r/i adenocarcinoma |
| Streaming | Absent | Absent | Present - "stretched yeast dough" | R/i squmaous (weak) |
| Keratin (difficult to see on Field stain) | Absent | Absent | Present | Present r/i squamous (strong) |
| Image | File:Lung small cell carcinoma -- extremely high mag.jpg SmCC - Pap stain. (WC) |
File:Lung adenocarcinoma - Pap stain -- very high mag.jpg LA - Pap stain. (WC) |
File:Squamous carcinoma - lung FNA -- very high mag.jpg SCC - Pap stain. (WC) |
Criteria list
Neuroendocrine tumours - look for:
- Nuclear moulding (not seen in NSCLC).
- Singular bare nuclei/single cells - often very abundant in small cell lung carcinoma (SCLC).
- Size ~2X neutrophil (PMN) - SCLC is large relative to most haematologic cancers (which are approx. the size of a PMN)... small in relation to other carcinoma.[6]
- Stippled chromatin.
- Negatives: Abundant cytoplasm - virtually excludes SCLC.
- Carcinoid vs. atypical carcinoid vs. SCLC (list from good to bad) - degree of nuclear atypia, presence of necrosis and smoking history.
- One should never sign-out small cell carcinoma without looking at the history.[7]
Adenocarcinoma:
- Nucleolus.
- Good ones are visible with 10X objective (excludes SCLC).
- Look for subtle large ones - at higher power.
- Neuroendocrine tumours occasionally may appear to have nucleoli - one should see good nucleoli in 3-4 cells in one field.
- Abundant cytoplasm - virtually excludes small cell carcinoma.
- Vacuoles with mucin (pink discolouration) - virtually diagnostic, though only seen occasionally.
- Eccentric nucleus.
- Negatives: NO moulding.
- Important if no nucleolus obvious.
Squamous cell carcinoma:
- Small nucleolus - not visible at 10X.
- Coarse chromatin.
- "Streaming" - think stringy yeast dough.
- Keratin (orange) - on Pap stain.
Adenocarcinoma
General
- Most common type of lung cancer.
Cytology
Features:
- Eccentric nucleus:
- +/-Nuclear grooves.
- +/-Nuclear pseudoinclusions.
- Nucleolus.
- Good ones are visible with 10X objective (virtually excludes SCLC).
- Look for subtle large ones - at higher power.
- Abundant cytoplasm - virtually excludes small cell carcinoma.
- Vacuoles with mucin (pink discolouration) - virtually diagnostic.
Notes:
- May be subtle, i.e. have minimal cytologic changes.
- No nuclear moulding -esp. important if no nucleolus visible.
DDx:
- Creola body - have cilia, may be subtle.
- Benign mesothelium (also sheets of cells).
- Atypical adenomatous hyperplasia (AAH) - thought to be the precursor to adenocarcinoma.[8]
- AAH has a size criterion, ergo not really possible to diagnose on cytopathology specimen.
- Vegetable cell - contaminant.[9]
Images
- Lung adenocarcinoma - Pap stain -- high mag.jpg
LA - Pap - high mag. (WC)
- Lung adenocarcinoma - Pap stain -- very high mag.jpg
LA - Pap - very high mag. (WC)
- Lung adenocarcinoma - Pap stain - alt -- very high mag.jpg
LA - Pap - very high mag. (WC)
LA - DQ - high mag. (WC)
LA - DQ - very high mag. (WC)
Neuroendocrine tumours
- This is a group of tumours that has benign (e.g. carcinoid tumour of the lung) to malignant (e.g. small cell lung carcinoma) behaviour.[10]
The grouping can be divided into four types:[11]
- Small cell carcinoma.
- Large cell neuroendocrine carcinoma.
- Typical carcinoid.
- Atypical carcinoid.
Cytologic features useful for differentiation:
- Small cell carcinoma: necrosis, scant cytoplasm, mitoses.
- Typical carcinoid: often more cytoplasm, no necrosis, low mitotic rate (MIB1: scant staining).
- Atypical carcinoid: higher mitotic rate/MIB1 than typical carcinoid,[12] no necrosis.
Notes:[11]
- Large cell and small cell tumours behave in a similar fashion; large cell can be considered a morphological variant of small cell.
- 9/10 of carcinoids are typical and usually have a good prognosis, i.e. do not metastasize.
- Central location (vis-a-vis peripheral location) tends favours typical carcinoid over atypical carcinoid.
Small cell lung carcinoma
General
- Is the most easy lung cancer to miss, as one is usually looking for large cells.
Microscopic
Features:
- Morphologic features of malignancy:
- Irregular nuclear membrane.
- Chromatin clumping.
- Marked nuclear size variation.
- Nuclear moulding - most useful for distinguishing from NSCLC[13][14] - key feature.
- Stippled chromatin - key feature.
- Scant cytoplasm - so scant it often near impossible to see - important feature.
- Small cells ~ 2x RBC.
- Bare nuclei common - very useful if present.
- Necrosis - essential.
- Cell fragmentation - smeared gray background (Pap stain), fragmented nuclei.
Notes:
- The Azzopardi phenomenon (smudging of nuclei) is not present on cytology specimens - it is processing artifact.
- Small cell carcinoma should not be diagnosed without a clinical history; if there is no smoking history... think about the possibility of carcinoid and atypical carcinoid.
- Small cell leukemias may mimic small cell carcinoma; difference: leukemias typically have smaller cells (~size of RBC vs. ~2x of RBC), and lymphoglandular bodies.
Image
Case 1
- Small cell lung cancer - cytology.jpg
SmCC showing nuclear moulding - Field stain. (WC)
Case 2
- Lung small cell carcinoma - Diff-Quik -- high mag.jpg
SmCC - Diff-Quik - high mag. (WC)
- Lung small cell carcinoma - Diff-Quik -- very high mag.jpg
SmCC - Diff-Quik - very high mag. (WC)
- Lung small cell carcinoma -- very high mag.jpg
SmCC - Pap stain - very high mag. (WC)
- Lung small cell carcinoma -- extremely high mag.jpg
SmCC - Pap stain - extremely high mag. (WC)
Case 3
SmCC - high mag.
SmCC - high mag.
- Small cell carcinoma - BRB - alt 2 -- high mag.jpg
SmCC - high mag.
- Small cell carcinoma - BRB -- very high mag.jpg
SmCC - very high mag.
- Small cell carcinoma - BRB - alt -- very high mag.jpg
SmCC - very high mag.
- Small cell carcinoma - BRB - alt 2 -- very high mag.jpg
SmCC - very high mag.
SmCC - high very mag.
Squamous cell carcinoma
Cytology
General - features:
- Nuclear features of malignancy (required):
- Irregular nuclear membrane, e.g. notches, sharp discontinuities.
- Nuclear hyperchromasia - "jet-black" nuclei on Pap stain key feature.
- Increased NC ratio.
- Variation of nuclear size from cell-to-cell.
Features - well-differentiated:
- Classically single cells.
- Keratinized = orange/red staining on Pap stain.
- "Intense" (blue) staining of cells on rapid Romanowsky + pyknotic (small shriveled) nucleus.[15]
Features - poorly differentiated:
- Mix of spindle cells/epithelioid cells usu. present in clusters without a common border.
- Usu. lack keratinization, i.e. not orange/red.
- "Dense" appearing cytoplasm.
- +/-Laminae (layers)/lines in the cytoplasm.
Notes:
- One should see abnormal squamous cells to call it SCC.
- One should think of adenocarcinoma as the default - it is more common.
- Poorly differentiated SCC may look like adenocarcinoma.
Image
Case 1
- Squamous carcinoma lung 1 cytology.jpg
SCC. (WC)
- Squamous carcinoma lung 2 cytology.jpg
SCC. (WC)
Case 2
- Squamous carcinoma - lung FNA -- high mag.jpg
SCC - high mag.
- Squamous carcinoma - lung FNA - alt -- high mag.jpg
SCC - high mag.
- Squamous carcinoma - lung FNA -- very high mag.jpg
SCC - very high mag.
- Squamous carcinoma - lung FNA - alt -- very high mag.jpg
SCC - very high mag.
- Squamous carcinoma - lung FNA -- high and very high mag - animation.gif
SCC animation - very high mag.
- Squamous carcinoma - lung FNA -- extremely high mag.jpg
SCC - extremely high mag.
- Squamous carcinoma - lung FNA - alt 2 -- extremely high mag.jpg
SCC - extremely high mag.
Malignant melanoma
Cytology
Classic features:
- Loosely cohesive cells and single cells.
- Mixure of epithelioid cells and spindle cells.
- Malignant cells have:
- Prominent red nucleolus.
- Pigmented cytoplasm - key feature (often not pigmented).
- Pigment may only be present in macrophages
- Occasional large binucleated cells (bug-eyed monster cell).
- Nuclei are often at opposite poles of the cell, i.e. the nuclei are as far apart as possible ("divorce cells").[16]
- Intranuclear inclusions.
- Pigmented macrophages (useful feature - but less specific for melanoma than pigment in malignant looking cells).
Notes:
- Large nucleolus - may Vaguely resemble adenocarcinoma.
- Prominent red nucleolus common in: serous carcinoma.
- The classic appearance of melanoma without pigment is closest to adenocarcinoma (which may have red nucleoli, large cells, abundant cytoplasm, occasional binucleation).
- Differentiating morphologic features: adenocarcinoma - 3-D clusters of cells, no spindle-shaped cells.
- Bug-eyed monster cells - may vaguely resemble a Reed-Sternberg cell (RSC) - diagnostic of Hodgkin's lymphoma (HL).
- RSCs do not have the granular cytoplasm typical of melanoma.
- Nuclei usually adjacent, i.e. not at opposite poles of the cell.
- Background of melanoma different than HL.
Images
- Melanoma - cytology field stain.jpg
Bug-eyed monster cell in melanoma. Field stain. (WC)
Other
Asthma
Cytology
Classic findings:
- Eosinophils.
- Curschmann's spirals = spiral-shaped mucous plugs.[17]
- Creola bodies = clusters of (ciliated) bronchial epithelial cells,[18] presence associated with more eosinophils.[19]
Note:
- Curschmann's spirals are a non-specific finding; they may be seen in a range of conditions in including chronic bronchitis, asymptomatic smokers and lung cancer.[17]
- They can also been in Pap tests.
Images
- Curshman's Spiral.jpg
Curschmann spiral. (WC)
www:
Pulmonary hamartoma
General
- Non-neoplastic growth - see hamartoma.
Microscopic
Features:
- No cytologic features of malignancy.
- Fat.
Drug-induced pulmonary toxicity
General
- Need clinical history.
Cytology
Features:
- Macrophages - classic for amiodarone toxicity.
Mucinous metaplasia
Cytology
Features:
- Cluster of cells with abundant clear cytoplasm.
- Bland nuclei.
DDx:
- Signet ring cell carcinoma - often single cells.
Lipoid pneumonia
General
Etiology:
- Aspiration or inhalation of a fatty substance, e.g. mineral oils (for constipation[21]), petroleum jelly.[22]
- Often does not have the classic associations seen in aspiration pneumonia, i.e. intoxication, neurologic disease.
Clinical:
- Chronic cough[23] - classic finding.
- +/-Dyspnea.[21]
- +/-Fever.
- Lower lobe air space disease - opacification (left>right).
- +/-Hemoptysis (uncommon).
Treatment:
- Stop exposure to lipoid material.[23]
DDx (clinical):
- Viral pneumonia.
- Others.
Cytology
Features:[22]
- Macrophages with multiple fat droplets.
- Often fill the cytoplasm.
- Nucleus often eccentric; not distorted (as seen in signet ring cells).
DDx:
- Signet ring cell carcinoma.
- Gastroesophageal reflux disease - also Oil red O +ve.[24]
Image
Stains
- Oil red O stain +ve.[25]
- Iron stain -ve.
Non-specific inflammation
- A very common finding in BALs.
Types:
- Eosinophilia
- If you notice eosinophils... you probably have eosinophilia.
- One in several HPFs (40x obj. with 22 mm eye piece) is enough.
- If you notice eosinophils... you probably have eosinophilia.
- Acute - neutrophils.
- ~10/HPF (40x obj. 22mm eye piece).
- Chronic - lymphocytes + occ. plasma cells.
- ~5 small lymphocytes/HPF (40x obj. 22 mm eye piece).
- Mixed acute & chronic inflammation.
See also
References
- ↑ Nayak, A.; Sugrue, C.; Koenig, S.; Wasserman, PG.; Hoda, S.; Morgenstern, NJ. (Feb 2012). "Endobronchial ultrasound-guided transbronchial needle aspirate (EBUS-TBNA): a proposal for on-site adequacy criteria.". Diagn Cytopathol 40 (2): 128-37. doi:10.1002/dc.21517. PMID 22246929.
- ↑ UHN PCY50001.08 P.11.
- ↑ URL: http://www.cytologystuff.com/study/section12ng.htm. Accessed on: 19 August 2015.
- ↑ URL: http://www.cytologystuff.com/study/section11ng.htm. Accessed on: 26 October 2015.
- ↑ Attributed to SM. 6 January 2010.
- ↑ WG. 20 January 2010.
- ↑ WG. 20 January 2010.
- ↑ Mori, M.; Rao, SK.; Popper, HH.; Cagle, PT.; Fraire, AE. (Feb 2001). "Atypical adenomatous hyperplasia of the lung: a probable forerunner in the development of adenocarcinoma of the lung.". Mod Pathol 14 (2): 72-84. doi:10.1038/modpathol.3880259. PMID 11235908. http://www.nature.com/modpathol/journal/v14/n2/full/3880259a.html.
- ↑ Naryshkin, S.; Young, NA. (1993). "Respiratory cytology: a review of non-neoplastic mimics of malignancy.". Diagn Cytopathol 9 (1): 89-97. PMID 8384547.
- ↑ URL: http://emedicine.medscape.com/article/426400-overview. Accessed on: 20 January 2010.
- ↑ 11.0 11.1 http://www.cancer.org/docroot/CRI/content/CRI_2_4_1X_What_is_lung_carcinoid_tumor_56.asp
- ↑ WG. February 2010.
- ↑ Sturgis, CD.; Nassar, DL.; D'Antonio, JA.; Raab, SS. (Aug 2000). "Cytologic features useful for distinguishing small cell from non-small cell carcinoma in bronchial brush and wash specimens.". Am J Clin Pathol 114 (2): 197-202. doi:10.1309/8MQG-6XEK-3X9L-A9XU. PMID 10941334. http://ajcp.ascpjournals.org/content/114/2/197.full.pdf.
- ↑ URL: http://onlinelibrary.wiley.com/doi/10.1002/dc.10297/abstract. Accessed on: 31 May 2011.
- ↑ GS. 24 February 2010.
- ↑ GS. 24 February 2010.
- ↑ 17.0 17.1 Cenci M, Giovagnoli MR, Alderisio M, Vecchione A (November 1998). "Curschmann's spirals in sputum of subjects exposed daily to urban environmental pollution". Diagn. Cytopathol. 19 (5): 349–51. PMID 9812228.
- ↑ Isohima, K.; Takahashi, K.; Soda, R.; Hukasaka, N.; Tanabe, K.; Ozaki, K.; Nakato, H.; Araki, M. et al. (Jul 1989). "[The clinical significance of Creola body in the sputum of asthmatic patients].". Arerugi 38 (7): 542-8. PMID 2818212.
- ↑ Motojima, S.; Kushima, A.; Ogata, H.; Tateishi, K.; Fukuda, T.; Makino, S. (Apr 1990). "[Relationship between presence of Creola bodies and airway hyperresponsiveness in patients with bronchial asthma].". Arerugi 39 (4): 377-83. PMID 2383179.
- ↑ URL: http://www.medeponyms.com/entry/27/. Accessed on: 31 March 2012.
- ↑ 21.0 21.1 Simmons, A.; Rouf, E.; Whittle, J. (Nov 2007). "Not your typical pneumonia: a case of exogenous lipoid pneumonia.". J Gen Intern Med 22 (11): 1613-6. doi:10.1007/s11606-007-0280-7. PMID 17846847.
- ↑ 22.0 22.1 22.2 Khilnani, GC.; Hadda, V. (Oct 2009). "Lipoid pneumonia: an uncommon entity.". Indian J Med Sci 63 (10): 474-80. doi:10.4103/0019-5359.57639. PMID 19901490.
- ↑ 23.0 23.1 Bell, MM. (Sep 2015). "Lipoid pneumonia: An unusual and preventable illness in elderly patients.". Can Fam Physician 61 (9): 775-7. PMID 26371101.
- ↑ Hopkins, PM.; Kermeen, F.; Duhig, E.; Fletcher, L.; Gradwell, J.; Whitfield, L.; Godinez, C.; Musk, M. et al. (Aug 2010). "Oil red O stain of alveolar macrophages is an effective screening test for gastroesophageal reflux disease in lung transplant recipients.". J Heart Lung Transplant 29 (8): 859-64. doi:10.1016/j.healun.2010.03.015. PMID 20466562.
- ↑ Yampara Guarachi, GI.; Barbosa Moreira, V.; Santos Ferreira, A.; Sias, SM.; Rodrigues, CC.; Teixeira, GH. (2014). "Lipoid pneumonia in a gas station attendant.". Case Rep Pulmonol 2014: 358761. doi:10.1155/2014/358761. PMID 25374742.