Adenocarcinoma of the lung
| Adenocarcinoma of the lung | |
|---|---|
| Diagnosis in short | |
|
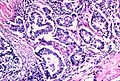
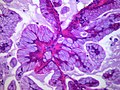
Template:Px Invasive adenocarcinoma, acinar pattern (right of image) and benign lung (left of image). H&E stain. | |
|
| |
| LM | +/-nuclear atypia (may be absent in mucinous tumours), eccentrically placed nuclei, usu. abundant cytoplasm (classically with mucin vacuoles), often conspicuous nucleoli, +/-nuclear pseudoinclusions |
| LM DDx | atypical adenomatous hyperplasia of the lung, adenocarcinoma in situ, squamous cell carcinoma of the lung, small cell carcinoma of the lung, non-small cell lung carcinoma, malignant mesothelioma, metastatic adenocarcinoma (esp. colorectal adenocarcinoma, breast adenocarcinoma (invasive ductal carcinoma of the breast, invasive lobular carcinoma)) |
| IHC | CK7 +ve, TTF-1 +ve, CK20 -ve, p40 -ve, p63 -ve (usually) |
| Molecular | +/-KRAS mutations, +/-EGFR mutations, +/-ALK chromosomal translocation (inv(2)(p21p23) -- EML4-ALK fusion), +/-ROS1 rearrangements, +/-RET rearrangements |
| Staging | lung cancer staging |
| Site | lung - see lung tumours |
|
| |
| Prevalence | most common primary lung tumour |
| Radiology | lung mass - typically peripheral lesion (distant from large airways), may be multifocal |
| Prognosis | dependent on stage (minimally invasive and noninvasive: very good; invasive: moderate) |
| Clin. DDx | other lung tumours - primary and metastatic |
| Treatment | surgical resection if feasible |
Adenocarcinoma of the lung, also lung adenocarcinoma, is common malignant lung tumour.
General
- Adenocarcinoma is the most common (primary lung cancer).[1]
- Adenocarcinoma is the non-smoker tumour - SCLC and squamous are more strongly associated with smoking.
- Lung adenocarcinoma is the most common brain metastasis.[2]
Treatment:
- Lung adenocarcinoma may be treated with EGFR inhibitors (e.g. gefitinib (Iressa), erlotinib (Tarceva)).[3]
Patients that receive EGFR inhibitors classically are:[4]
- Non-smokers.
- Female.
- Asian.
- Caucasians also benefit.[5]
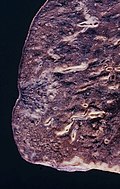
Gross
- Classically peripheral lesions.
- May be multifocal.
Image
Microscopic
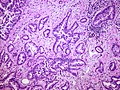
Features:
- +/-Nuclear atypia - important.
- May be absent in mucinous tumours - may look similar to foveolar epithelium.
- Eccentrically placed nuclei.
- Abundant cytoplasm - classically with mucin vacuoles.
- Often conspicuous nucleoli.
- +/-Nuclear pseudoinclusions.
Negatives:
- Lack of intercellular bridges.
Patterns:[6]
- Lepidic - tumour grows long the alveolar wall; means scaly covering.[7] At lower power, the shapes should still resemble lung acini.
- Acinar - berry-shaped glands, smaller than lung acini.
- Papillary - fibrovascular cores.
- Micropapillary - nipple shaped projections without fibrovascular cores.
- Solid - sheet of cells.
Notes:
- Lymphovascular invasion is common.
- Micropapillary predominant pattern and tumours with any amount of the lepidic pattern are associated with EGFR mutations.[8]
DDx:
- Atypical adenomatous hyperplasia of the lung - spaced hobnail cells, mild-to-moderate nuclear atypia, small lesion (must be <5 mm).
- Adenocarcinoma in situ.
- Papillary carcinoma of thyroid.
- Squamous cell carcinoma of the lung.
- Small cell carcinoma of the lung.
- Adenoid cystic carcinoma.
- Non-small cell lung carcinoma - diagnosis should be avoided if possible.
- Malignant mesothelioma.
- Metastatic adenocarcinoma.
- Colorectal adenocarcinoma.
- Breast adenocarcinoma.
- Other carcinomas.
- Carcinomas of the bronchial glands, e.g. adenoid cystic carcinoma.
Images
Acinar adenocarcinoma
Acinar LA - low mag.
Acinar LA - intermed. mag.
Acinar LA - high mag.
Acinar LA - very high mag.
Acinar LA - very high mag.
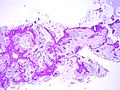
Mucinous adenocarcinoma
- Mucinous adenocarcinoma of the lung -- low mag.jpg
MAL - low mag.
- Mucinous adenocarcinoma of the lung -- intermed mag.jpg
MAL - intermed. mag.
- Mucinous adenocarcinoma of the lung -- high mag.jpg
MAL - high mag.
- Mucinous adenocarcinoma of the lung -- very high mag.jpg
MAL - very high mag.
- Mucinous lung adenocarcinoma -- low mag.jpg
MAL - low mag.
- Mucinous lung adenocarcinoma -- intermed mag.jpg
MAL - intermed. mag.
- Mucinous lung adenocarcinoma -- high mag.jpg
MAL - high mag.
- Mucinous lung adenocarcinoma and airway -- intermed mag.jpg
MAL - intermed. mag.
- Mucinous lung adenocarcinoma and airway -- high mag.jpg
MAL - high mag.
- Mucinous lung adenocarcinoma and airway - alt -- high mag.jpg
MAL - high mag.
Papillary adenocarcinoma
- Papillary adenocarcinoma of the lung -- very low mag.jpg
PAL - very low mag. (WC/Nephron)
- Papillary adenocarcinoma of the lung -- low mag.jpg
PAL - low mag. (WC/Nephron)
- Papillary adenocarcinoma of the lung -- intermed mag.jpg
PAL - intermed. mag. (WC/Nephron)
PAL - high mag. (WC/Nephron)
Fetal adenocarcinoma
- Fetal adenocarcinoma of the lung -- very low mag.jpg
FAL - very low mag. (WC)
- Fetal adenocarcinoma of the lung - alt2 -- very low mag.jpg
FAL - very low mag. (WC)
- Fetal adenocarcinoma of the lung -- low mag.jpg
FAL - low mag. (WC)
- Fetal adenocarcinoma of the lung -- intermed mag.jpg
FAL - intermed. mag. (WC)
- Fetal adenocarcinoma of the lung -- high mag.jpg
FAL - high mag. (WC)
www
- BAC mucinous type adjacent to benign (pathpedia.com).
- BAC mucinous and nonmucinous (cancergrace.org).[9]
- Lepidic adenocarcinoma with invasive (flickr.com/Yale Rosen).
- Lepidic adenocarcinoma (flickr.com/Yale Rosen).
- Lepidic adenocarcinoma (rosaicollection.org/index.cfm).
- Mucinous adenocarcinoma (pathlabmed.typepad.com).
- Non-mucinous adenocarcinoma in situ (flickr.com/Yale Rosen).
- Non-mucinous adenocarcinoma in situ (flickr.com/Yale Rosen).
- Acinar adenocarcinoma (flickr.com/Yale Rosen).
- Acinar adenocarcinoma (flickr.com/Yale Rosen).
Classification
Classification based on extent:[6]
- Adenocarcinoma in situ (AIS) - previously known as bronchioloalveolar carcinoma (abbreviated BAC).
- Subtypes: nonmucinous, mucinous, mixed mucinous/nonmucinous.
- Definition: lack of invasion into the stroma, vascular spaces and pleura.
- Must have a lepidic growth pattern.[10]
- Minimally invasive adenocarcinoma (MIA).
- Lepidic growth with up to 5 mm of invasion.
- Subtypes: nonmucinous (most common), mucinous (uncommon), mixed (mucinous/nonmucinous).
- Should not have lymphovascular invasion.[citation needed]
- Invasive adenocarcinoma:
- Subtypes: micropapillary, mucinous (previously mucinous BAC), colloid, fetal, enteric.
Grading
Graded G1-G4 - as per CAP protocol (version 3.4.0.0):[11]
- G1 = lepidic.
- G2 = acinar, papillary, cribriform.
- G3 = micropapillary, solid, mucinous, colloid.
- G4 = undifferentiated - not used for lung adenocarcinoma; it used for small cell carcinoma and large cell carcinoma.
Note:
- There is no consensus currently on grading - as per the international consensus guidelines of 2011.[6]
Special stains
- Mucicarmine +ve, cytoplasmic.
- PAS-diastase +ve, cytoplasmic.
IHC
Primary versus metastatic:
- TTF-1 +ve.
- CK7 +ve.
- CK20 -ve.
Panel for adenocarcinoma versus SCC:
Others:
- p63 -ve -- occasionally +ve.
- Vimentin -ve/+ve (+ve relatively common).
- Poor prognosticator.[13]
Note:
- In mucinous adenocarcinoma of the lung TTF-1 is usu. -ve (46% +ve) and napsin is usu. -ve (36% +ve).
- Positive staining is unusual but useful if present, as metastatic disease is uniformily negative for both.[14]
Molecular
- EGFR mutations (typically assessed by PCR) - respond to TKIs (e.g. gefitinib, erlotinib) if:[15]
- Exon 19 deletion.
- Exon 21 L858R.
- Natural history of mutation is suspected to have a better prognosis vs. wild-type.[16]
- KRAS mutations are absent, i.e. wild-type KRAS.[17]
- ALK chromosomal translocation (inv(2)(p21p23) -- EML4-ALK fusion).[18]
- Associated with a poor prognosis.[19]
- Amenable to treatment with TKI.
- See lung carcinoma with ALK rearrangement.
- Do not occur with EGRF mutations or KRAS mutations.[20]
Sign out
Biopsy
Consensus recommendations:[6]
- Adenocarcinoma in situ (AIS) and minimally invasive adenocarcinoma should not be used in the reporting of small biopsies and cytology.
- Tumours with a non-invasive pattern are referred to by their pattern, e.g. lepidic growth, not as AIS.
Lung, Right Upper Lobe, Core Biopsy: - INVASIVE ADENOCARCINOMA, NON-MUCINOUS. Comment: The adenocarcinoma is positive for TTF-1 and napsin. EGFR/ALK testing was ordered.
Lepidic pattern on biopsy
Important note: lesion must be >=5 mm and <=30 mm.
Lung, Left Upper Lobe, Core Biopsy:
- ADENOCARCINOMA, lepidic pattern.
Comment:
The tumour is stains as follows:
POSITIVE: TTF-1, napsin A, CK7.
NEGATIVE: p40.
The findings are in keeping with lepidic pattern adenocarcinoma; the differential diagnosis includes: (1) adenocarcinoma in situ, (2) minimally invasive adenocarcinoma, and (3) invasive adenocarcinoma.
Lung biomarkers (EGFR, ALK, PDL1, ROS1) have been ordered.
Mucinous adenocarcinoma with noncontributory stains
Lung, Right Upper Lobe, Core Biopsy: - ADENOCARCINOMA, MUCINOUS, see comment. Comment: The adenocarcinoma is negative for both napsin and TTF-1. EGFR/ALK testing was ordered. The findings are compatible with a primary or secondary adenocarcinoma; clinical and radiologic correlation is required.
Block letters
LUNG, LEFT, BIOPSY: - ADENOCARCINOMA, LEPIDIC GROWTH; INVASION CANNOT BE EXCLUDED IN THIS SMALL SPECIMEN.
LUNG, RIGHT UPPER LOBE, NEEDLE BIOPSY: - INVASIVE ADENOCARCINOMA, NON-MUCINOUS. COMMENT: The tumour stains as follows: POSITIVE: TTF-1. NEGATIVE: p40. The immunoprofile is compatible with lung adenocarcinoma.
MASS, LEFT LOWER LOBE OF LUNG, BIOPSY: - INVASIVE ADENOCARCINOMA. COMMENT: The tumour is positive for TTF-1. Tissue will be sent for molecular testing and the results reported as an addendum.
Resection
LUNG, LEFT UPPER LOBE, LOBECTOMY: - ADENOCARCINOMA WITH AN ACINAR PATTERN, SOLID PATTERN, MICROPAPILLARY PATTERN AND LEPIDIC PATTERN -- PATTERNS IN ORDER OF PREVALENCE. - MARGINS NEGATIVE FOR MALIGNANCY. - THREE LYMPH NODES NEGATIVE FOR MALIGNANCY (0 POSITIVE/3). - PLEASE SEE TUMOUR SUMMARY.
LUNG, RIGHT UPPER LOBE, LOBECTOMY: - MULTIPLE ADENOCARCINOMAS (x2) WITH AN ACINAR PATTERN, SOLID PATTERN, MICROPAPILLARY PATTERN AND LEPIDIC PATTERN -- PATTERNS IN ORDER OF PREVALENCE. - MARGINS NEGATIVE FOR MALIGNANCY. - FOUR LYMPH NODES NEGATIVE FOR MALIGNANCY (0 POSITIVE/4). - LYMPHOVASCULAR INVASION PRESENT. - PLEASE SEE TUMOUR SUMMARY AND COMMENT. COMMENT: The histology of the two adenocarcinomas resemble one another and lymphovascular invasion is present. These findings favour that the smaller tumor is a metastasis, rather than a synchronous primary.
Micro
Size (tissue): scant tissue (<0.5 cm). Gland formation: focal, poorly formed. Cell size: large. Cytoplasm: moderate-to-abundant, grey-eosinophilic. Nucleus location: eccentric. Nuclear pleomorphism: moderate. Nuclear moulding: absent. Nucleoli: present, prominent. Nuclear pseudoinclusions: present.
Number of cores: 3. Length of cores (total): 2.0 cm. Gland formation: present. Cell size: large. Cytoplasm: moderate, grey-eosinophilic. Necrosis: none apparent. Mucin: none. Nucleus location: eccentric. Nuclear pleomorphism: moderate. Nuclear moulding: absent. Nuclear pseudoinclusions: absent. Nuclear shape/arrangment: cigar-like/pseudostratified. Nucleoli: present.
Mucinous
The sections show cores with well-formed glands composed of foveolar-like columnar cells with a relatively bland cytomorphology. Mitotic activity is not readily apparent. A small amount of non-lesional lung parenchyma is present.
Lung cancer staging
See also
- Lung tumours.
- Adenocarcinoma.
- Metastasis.
- Lung carcinoma with ALK rearrangement.
- SMARCA4-deficient adenocarcinoma of the lung.
References
- ↑ Lutschg JH (January 2009). "Lung cancer". N. Engl. J. Med. 360 (1): 87-8; author reply 88. doi:10.1056/NEJMc082208. PMID 19118313.
- ↑ Nayak, L.; Lee, EQ.; Wen, PY. (Feb 2012). "Epidemiology of brain metastases.". Curr Oncol Rep 14 (1): 48-54. doi:10.1007/s11912-011-0203-y. PMID 22012633.
- ↑ Sun Y, Ren Y, Fang Z, et al. (October 2010). "Lung adenocarcinoma from East Asian never-smokers is a disease largely defined by targetable oncogenic mutant kinases". J. Clin. Oncol. 28 (30): 4616–20. doi:10.1200/JCO.2010.29.6038. PMID 20855837.
- ↑ Job B, Bernheim A, Beau-Faller M, et al. (2010). "Genomic Aberrations in Lung Adenocarcinoma in Never Smokers". PLoS One 5 (12): e15145. doi:10.1371/journal.pone.0015145. PMC 2997777. PMID 21151896. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2997777/.
- ↑ Rosell, R.; Moran, T.; Cardenal, F.; Porta, R.; Viteri, S.; Molina, MA.; Benlloch, S.; Taron, M. (Oct 2010). "Predictive biomarkers in the management of EGFR mutant lung cancer.". Ann N Y Acad Sci 1210: 45-52. doi:10.1111/j.1749-6632.2010.05775.x. PMID 20973798.
- ↑ 6.0 6.1 6.2 6.3 Travis WD, Brambilla E, Noguchi M, et al. (February 2011). "International association for the study of lung cancer/american thoracic society/european respiratory society international multidisciplinary classification of lung adenocarcinoma". J Thorac Oncol 6 (2): 244–85. doi:10.1097/JTO.0b013e318206a221. PMID 21252716.
- ↑ URL: http://medical-dictionary.thefreedictionary.com/lepidic. Accessed on: 8 August 2013.
- ↑ Shim, HS.; Lee, da H.; Park, EJ.; Kim, SH. (Oct 2011). "Histopathologic characteristics of lung adenocarcinomas with epidermal growth factor receptor mutations in the International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society lung adenocarcinoma classification.". Arch Pathol Lab Med 135 (10): 1329-34. doi:10.5858/arpa.2010-0493-OA. PMID 21970488.
- ↑ URL: http://cancergrace.org/lung/2007/05/14/bac-mucinous-and-non-mucinous/. Accessed on: 8 August 2013.
- ↑ Borczuk, AC. (Jan 2012). "Assessment of invasion in lung adenocarcinoma classification, including adenocarcinoma in situ and minimally invasive adenocarcinoma.". Mod Pathol 25 Suppl 1: S1-10. doi:10.1038/modpathol.2011.151. PMID 22214965.
- ↑ CAP Lung protocol. Version: 3.4.0.0. URL: http://www.cap.org/ShowProperty?nodePath=/UCMCon/Contribution%20Folders/WebContent/pdf/cp-lung-16protocol-3400.pdf. Accessed on: March 23, 2016.
- ↑ Bishop, JA.; Teruya-Feldstein, J.; Westra, WH.; Pelosi, G.; Travis, WD.; Rekhtman, N. (Mar 2012). "p40 (ΔNp63) is superior to p63 for the diagnosis of pulmonary squamous cell carcinoma.". Mod Pathol 25 (3): 405-15. doi:10.1038/modpathol.2011.173. PMID 22056955.
- ↑ Dauphin, M.; Barbe, C.; Lemaire, S.; Nawrocki-Raby, B.; Lagonotte, E.; Delepine, G.; Birembaut, P.; Gilles, C. et al. (Jul 2013). "Vimentin expression predicts the occurrence of metastases in non small cell lung carcinomas.". Lung Cancer 81 (1): 117-22. doi:10.1016/j.lungcan.2013.03.011. PMID 23562674.
- ↑ Rossi, G.; Cavazza, A.; Righi, L.; Sartori, G.; Bisagni, A.; Longo, L.; Pelosi, G.; Papotti, M. (Aug 2014). "Napsin-A, TTF-1, EGFR, and ALK Status Determination in Lung Primary and Metastatic Mucin-Producing Adenocarcinomas.". Int J Surg Pathol 22 (5): 401-7. doi:10.1177/1066896914527609. PMID 24651909.
- ↑ John, T.; Liu, G.; Tsao, MS. (Aug 2009). "Overview of molecular testing in non-small-cell lung cancer: mutational analysis, gene copy number, protein expression and other biomarkers of EGFR for the prediction of response to tyrosine kinase inhibitors.". Oncogene 28 Suppl 1: S14-23. doi:10.1038/onc.2009.197. PMID 19680292.
- ↑ URL: http://www.mycancergenome.org/mutation.php?dz=nsclc&gene=egfr&code=l858r. Accessed on: 27 April 2012.
- ↑ Pao, W.; Wang, TY.; Riely, GJ.; Miller, VA.; Pan, Q.; Ladanyi, M.; Zakowski, MF.; Heelan, RT. et al. (Jan 2005). "KRAS mutations and primary resistance of lung adenocarcinomas to gefitinib or erlotinib.". PLoS Med 2 (1): e17. doi:10.1371/journal.pmed.0020017. PMID 15696205.
- ↑ Li, Y.; Ye, X.; Liu, J.; Zha, J.; Pei, L. (Jan 2011). "Evaluation of EML4-ALK fusion proteins in non-small cell lung cancer using small molecule inhibitors.". Neoplasia 13 (1): 1-11. PMID 21245935.
- ↑ Yang, P.; Kulig, K.; Boland, JM.; Erickson-Johnson, MR.; Oliveira, AM.; Wampfler, J.; Jatoi, A.; Deschamps, C. et al. (Jan 2012). "Worse disease-free survival in never-smokers with ALK+ lung adenocarcinoma.". J Thorac Oncol 7 (1): 90-7. doi:10.1097/JTO.0b013e31823c5c32. PMID 22134072.
- ↑ Gainor, JF.; Varghese, AM.; Ou, SH.; Kabraji, S.; Awad, MM.; Katayama, R.; Pawlak, A.; Mino-Kenudson, M. et al. (Aug 2013). "ALK rearrangements are mutually exclusive with mutations in EGFR or KRAS: an analysis of 1,683 patients with non-small cell lung cancer.". Clin Cancer Res 19 (15): 4273-81. doi:10.1158/1078-0432.CCR-13-0318. PMID 23729361.
- ↑ Shaw, AT.; Ou, SH.; Bang, YJ.; Camidge, DR.; Solomon, BJ.; Salgia, R.; Riely, GJ.; Varella-Garcia, M. et al. (Nov 2014). "Crizotinib in ROS1-rearranged non-small-cell lung cancer.". N Engl J Med 371 (21): 1963-71. doi:10.1056/NEJMoa1406766. PMID 25264305.
- ↑ Gold, KA. (Nov 2014). "ROS1--targeting the one percent in lung cancer.". N Engl J Med 371 (21): 2030-1. doi:10.1056/NEJMe1411319. PMID 25409376.