Gastrointestinal stromal tumour
Jump to navigation
Jump to search
The printable version is no longer supported and may have rendering errors. Please update your browser bookmarks and please use the default browser print function instead.
| Gastrointestinal stromal tumour | |
|---|---|
| Diagnosis in short | |
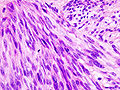
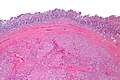
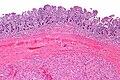
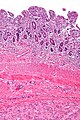
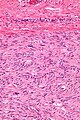
 Gastrointestinal stromal tumour. H&E stain. | |
|
| |
| LM | spindle or epithelioid or mixed morphology, usu. centred on the muscularis propria |
| LM DDx | schwannoma, leiomyoma, leiomyosarcoma, neurofibroma, desmoid-type fibromatosis |
| IHC | CD117 +ve, DOG1 +ve, CD34 +ve, S-100 -ve |
| Molecular | mutation in KIT gene or PDGFRA gene |
| Staging | gastrointestinal stromal tumour staging |
| Site | stomach, small intestine, other sites |
|
| |
| Syndromes | Neurofibromatosis type 1, Carney triad, Carney-Stratakis syndrome |
|
| |
| Prognosis | good to poor - dependent on size, site & mitotic rate |
The gastrointestinal stromal tumour, abbreviated GIST, is an uncommon tumour of the gastrointestinal tract.
General
Definition
- Tumour resulting from a mutation in the KIT gene or PDGFRA (Platelet-derived growth factor receptor, alpha polypeptide) gene.[1]
- Cases wild-type for KIT or PDFGRA may harbour defects in the succinate dehydrogenase complex, NF-1, BRAF, or extremely rarely KRAS.
Epidemiology
- Arise from Interstitial cells of Cajal.[1]
May be familial/syndromic:[2]
- Neurofibromatosis 1 (von Recklinghausen's disease).
- Carney triad.
- Carney-Stratakis syndrome - GISTs and paraganglioma - due to mutation in the genes for succinate dehydrogenase.[3]
Treatment
- Imatinib (Gleevec) - drug was developed for chronic myelogenous leukemia.
Factors predictive of malignant behaviour
Features suggesting a bad prognosis:[1]
- Large size.
- Often benign if small size.
- High mitotic rate (for area 5mm2).
- Site - small intestine GISTs worse than stomach GISTs.
Small intestine bad prognosis:[1]
- >5 mitoses/5 mm2 or size >10 cm.
Stomach bad prognosis:[1]
- >5 mitoses/5 mm2 and size >5 cm.
Location
Most common locations in order:[1]
- 60% in stomach.
- 35% in small intestine.
- 5% elsewhere.
Notes:
- Small intestinal GISTs have a worse prognosis than gastric ones.[1]
- GISTs almost never metastasize to the lymph nodes (except for SDH-B deficient epithelioid GISTs)
- Most common metastasis locations: liver, abdominal soft tissue.
Microscopic
Features:
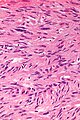
- Classically, spindle cell morphology ~ 50% of tumours.[4]
- May be epithelioid (round) ~40% of tumours.
- Mixed epithelioid and spindle cell tumours ~10% tumours.
- +/-Cytoplasmic inclusions[5] - perinuclear.[6]
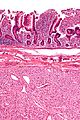
- Classically splits the layers of the muscularis propria - as this is where the interstitial cells of Cajal are located.[7]
- +/-Skenoid fibres - extracellular collagen bundles[8] ~ 2-5 x 60 micrometers - uncommon finding.
- Not seen in gastric GISTs.[9]
- High specificity for GIST.
DDx
- Leiomyosarcoma.
- Leiomyoma - esp. in the esophagus.
- Neural tumours.
- Neurofibroma.
- Schwannoma (GFAP +ve).
- GFAP uniformly neg. in GISTs.[1]
- Desmoid-type fibromatosis.
- Epstein-Barr virus-associated smooth muscle tumour - very uncommon, in immunoincompetent individuals.[10]
Images
- www:
IHC
- CD117 +ve in 95%.[1]
- Mast cells are the internal positive control.
- DOG1 +ve.[11]
Others:
IHC work-up panel
- S-100 (neural tumours, rarely +ve in GISTs[1]).
- CD34, CD117 (GIST).
- Desmin (muscle tumours).
Molecular tests
- Sequence Kit gene, PDGFRA gene.
Gastrointestinal stromal tumour staging
Main article: Gastrointestinal stromal tumour staging
GIST has its own staging.
Sign out
STOMACH (MASS), LESSER CURVE, WEDGE RESECTION: - GASTROINTESTINAL STROMAL TUMOUR (GIST). -- MARGINS NEGATIVE FOR GIST. COMMENT: The tumour stains as follows: POSITIVE: CD117, CD34. NEGATIVE: Desmin, S-100.
SMALL BOWEL (ILEUM), RESECTION: - GASTROINTESTINAL STROMAL TUMOUR (GIST), LOW-GRADE, NO RISK OF PROGRESSIVE DISEASE. -- MARGINS NEGATIVE FOR GIST. -- PLEASE SEE TUMOUR SUMMARY. - THREE BENIGN LYMPH NODES. COMMENT: The tumour stains as follows: POSITIVE: CD117, CD34. NEGATIVE: Desmin, S-100. PROLIFERATION (Ki-67): <1%.
Incidental GIST
Partial Stomach, Sleeve Gastrectomy: - Stomach wall with incidental GASTROINTESTINAL STROMAL TUMOUR (GIST), 2 mm in maximal dimension. -- Margin clear. - Gastric mucosa within normal limits. Comment: The tumour stains as follows: POSITIVE: DOG1, CD117, CD34. NEGATIVE: desmin, S-100. PROLIFERATION (Ki-67): <2%.
Staging
- The stage is primarily determined by the tumour size and mitotic grade.
- In the stomach, the mitotic grade determines whether a given tumour is Stage I or Stage III.[15]
Micro
The sections show a spindle cell lesion that is well-circumscribed and without significant nuclear pleomorphism. No lymphocytic cuff is surrounding the lesion. The lesion is focally seen at the inked soft tissue margin. Three mitoses are seen in 5 mm*mm.
See also
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 Miettinen M, Lasota J (October 2006). "Gastrointestinal stromal tumors: review on morphology, molecular pathology, prognosis, and differential diagnosis". Arch. Pathol. Lab. Med. 130 (10): 1466–78. PMID 17090188. http://journals.allenpress.com/jrnlserv/?request=get-abstract&issn=0003-9985&volume=130&page=1466.
- ↑ Agaimy A, Hartmann A (October 2010). "[Hereditary and non-hereditary syndromic gastointestinal stromal tumours]" (in German). Pathologe 31 (6): 430–7. doi:10.1007/s00292-010-1354-6. PMID 20848108.
- ↑ Blay, JY.; Blomqvist, C.; Bonvalot, S.; Boukovinas, I.; Casali, PG.; De Alava, E.; Dei Tos, AP.; Dirksen, U. et al. (Oct 2012). "Gastrointestinal stromal tumors: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up.". Ann Oncol 23 Suppl 7: vii49-55. doi:10.1093/annonc/mds252. PMID 22997454. http://annonc.oxfordjournals.org/content/23/suppl_7/vii49.full.
- ↑ Miettinen, M.; Sobin, LH.; Lasota, J. (Jan 2005). "Gastrointestinal stromal tumors of the stomach: a clinicopathologic, immunohistochemical, and molecular genetic study of 1765 cases with long-term follow-up.". Am J Surg Pathol 29 (1): 52-68. PMID 15613856.
- ↑ Pasquinelli G, Severi B, Martinelli GN, Santini D, Gelli MC, Tison V (April 1995). "Gastro-intestinal stromal tumors: an ultrastructural reinterpretation of the clear cell component". J. Submicrosc. Cytol. Pathol. 27 (2): 251–7. PMID 7757951.
- ↑ Boşoteanu, M.; Boşoteanu, C.; Deacu, M.; Aşchie, M. (2011). "Differential diagnosis of a gastric stromal tumor: case report and literature review.". Rom J Morphol Embryol 52 (4): 1361-8. PMID 22203947.
- ↑ Agaimy A, Wünsch PH (August 2006). "Gastrointestinal stromal tumours: a regular origin in the muscularis propria, but an extremely diverse gross presentation. A review of 200 cases to critically re-evaluate the concept of so-called extra-gastrointestinal stromal tumours". Langenbecks Arch Surg 391 (4): 322–9. doi:10.1007/s00423-005-0005-5. PMID 16402273.
- ↑ 8.0 8.1 8.2 Levy, AD.; Patel, N.; Dow, N.; Abbott, RM.; Miettinen, M.; Sobin, LH.. "From the archives of the AFIP: abdominal neoplasms in patients with neurofibromatosis type 1: radiologic-pathologic correlation.". Radiographics 25 (2): 455-80. doi:10.1148/rg.252045176. PMID 15798063.
- ↑ 9.0 9.1 Greenson, JK. (Apr 2003). "Gastrointestinal stromal tumors and other mesenchymal lesions of the gut.". Mod Pathol 16 (4): 366-75. doi:10.1097/01.MP.0000062860.60390.C7. PMID 12692202.
- ↑ Deyrup, AT.; Lee, VK.; Hill, CE.; Cheuk, W.; Toh, HC.; Kesavan, S.; Chan, EW.; Weiss, SW. (Jan 2006). "Epstein-Barr virus-associated smooth muscle tumors are distinctive mesenchymal tumors reflecting multiple infection events: a clinicopathologic and molecular analysis of 29 tumors from 19 patients.". Am J Surg Pathol 30 (1): 75-82. PMID 16330945.
- ↑ Liegl, B.; Hornick, JL.; Corless, CL.; Fletcher, CD. (Mar 2009). "Monoclonal antibody DOG1.1 shows higher sensitivity than KIT in the diagnosis of gastrointestinal stromal tumors, including unusual subtypes.". Am J Surg Pathol 33 (3): 437-46. doi:10.1097/PAS.0b013e318186b158. PMID 19011564.
- ↑ Bing, Z.; Pasha, TL.; Acs, G.; Zhang, PJ. (Jul 2008). "Cytoplasmic overexpression of WT-1 in gastrointestinal stromal tumor and other soft tissue tumors.". Appl Immunohistochem Mol Morphol 16 (4): 316-21. doi:10.1097/PAI.0b013e31815c2e02. PMID 18528287.
- ↑ Lasota, J.; Jasinski, M.; Sarlomo-Rikala, M.; Miettinen, M. (Jan 1999). "Mutations in exon 11 of c-Kit occur preferentially in malignant versus benign gastrointestinal stromal tumors and do not occur in leiomyomas or leiomyosarcomas.". Am J Pathol 154 (1): 53-60. doi:10.1016/S0002-9440(10)65250-9. PMID 9916918.
- ↑ Wada, N.; Kurokawa, Y.; Takahashi, T.; Hamakawa, T.; Hirota, S.; Naka, T.; Miyazaki, Y.; Makino, T. et al. (2016). "Detecting Secondary C-KIT Mutations in the Peripheral Blood of Patients with Imatinib-Resistant Gastrointestinal Stromal Tumor.". Oncology 90 (2): 112-7. doi:10.1159/000442948. PMID 26779618.
- ↑ Coccolini, F.; Catena, F.; Ansaloni, L.; Pinna, AD. (Feb 2012). "Gastrointestinal stromal tumor and mitosis, pay attention.". World J Gastroenterol 18 (6): 587-8. doi:10.3748/wjg.v18.i6.587. PMID 22363128.